Introduction
Vascular endothelial growth factor (VEGF), also known as vascular permeability factor (VPF), is a highly specific vascular endothelial cell growth factor that promotes increased vascular permeability and extracellular Stromal degeneration, vascular endothelial cell migration, proliferation, and angiogenesis. Among them, VEGF-A plays an important role in the regeneration of blood vessels and endothelial cells. In addition, in the embryonic stage, the role of VEGF is to regulate the proliferation, metastasis of embryonic cells and improve the survival rate of endothelial cells, and to increase the efficiency of bone formation through the recruitment of osteoblasts and chondroblasts. Moreover, VEGF is involved in the onset and progression of many angiogenesis-dependent diseases, including cancer, certain inflammatory diseases, and diabetic retinopathy.
Mechanism and Function
VEGF belongs to the platelet derived growth factor (PDGF) factor family, including VEGF-A, VEGF-B, VEGF-C, VEGF-D, VEGF-E, and placental growth factor (PGF). VEGF-A can promote neovascularization and increase vascular permeability. VEGF-B plays a role in non-angiogenic tumors. VEGF-C and VEGF-D play a role in the formation of neovascular and lymphatic vessels in cancer tissues. VEGF-E is also a potential neovascularization factor. PGF promotes neovascularization, increases vascular permeability, and significantly increases the expression of PGF in experimental choroidal neovascularization. High-affinity receptors that specifically bind to VEGF are called vascular endothelial growth factor receptors (VEGFRs), and are mainly divided into 3 types of VEGFR-1, VEGFR-2, and VEGFR-3. VEGFR-1 and VEGFR-2 are mainly distributed on the surface of tumor vascular endothelial cells and regulate the formation of tumor blood vessels. VEGFR-3 are mainly distributed on the surface of lymphatic endothelial cells and regulate the generation of tumor lymphatic vessels.
As a specific mitogen of vascular endothelial cells, VEGF can promote the growth of vascular endothelial cells in vitro and induce vascular proliferation in vivo. Especially in the hypoxic environment, VEGF binds to the VEGFR on the endothelial cell membrane, causing phosphorylation of the receptor, binding of the phosphorylated receptor to SHC, causing SHC phosphorylation, and activating SHC binding to the linker protein (GRB2) . There are two SH3 domains in Grb2, which can bind to guanine nucleotide release factor called SOS and change its activity. Then, SOS further activates Ras. Activated Ras acts on mitogen-activated protein kinase (MAPK) and activates ERK. Finally, ERK transfers into the nucleus, causing changes in the activity of certain transcription factors to achieve the mitogen properties of VEGF and induce endothelial cell proliferation.
In addition to promoting endothelial cell proliferation, VEGF also has the following three biological functions. First, promote angiogenesis. Under hypoxic environment, VEGF can increase the activity of plasma zymogen activator and promote extracellular protein by increasing the mRNA expression of plasma zymogen activator (PA) and Hydrolysis promotes the formation of new capillaries. Second, increase vascular permeability. It is characterized by rapid action and short duration. Third, change the extracellular matrix. Under hypoxic environment, VEGF can induce plasma prolysin activator and plasma lysogen activator inhibitor-1, as well as matrix collagenase, induce tissue factor expression in endothelial cells, and stimulate factor V3 from endothelial cells. Released, thereby altering the extracellular matrix and making it easier for blood vessels to grow.
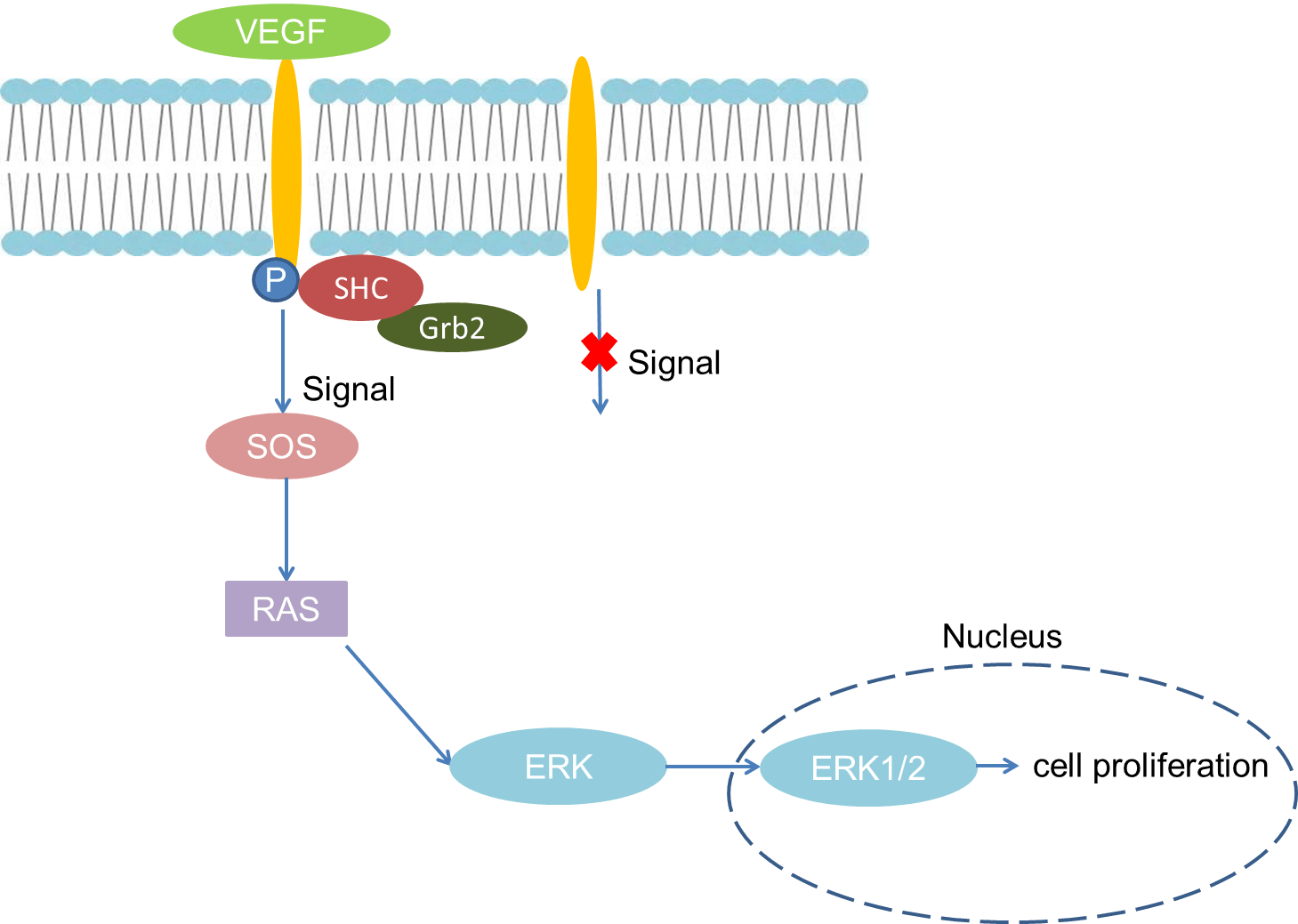 Fig 1. Mechanism of Signaling
Fig 1. Mechanism of Signaling
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- One or more cytokines cytokines qualitative and quantitative detection
- Cytokines qualitative and quantitative detection of various species
- Cytokine antibodies qualitative and quantitative detection
Sample requirements
- Sample Types-Bone tissue, heart muscle, liver parenchyma, osteoblasts, neutrophils, macrophages, keratinocytes, brown adipose tissue, etc.
- Sample Volume - It is optimal for 50 samples. This volume allows for triplicate testing of each sample.
Our advantages:
- Different detection methods can be selected based on different samples and requirements.
- Ensure the specificity and accuracy of the test by using high quality antibodies.
- Repeat the test to ensure the repeatability and accuracy of the experimental results.
- Feedback results are accurate and efficient
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow
For more information about the VEGF detection service or need other detection requirements, please contact us.
References:
- Bates DO, Heald RI, et al. Vascular endothelial growth factor increases Rana vascular permeability and compliance by different signaling pathways. J Physiol,2001,533(Pt 1):263-272.
- Shi W, Siemann DW. Inhibition of renal cell carcinoma angiogenesis and growth by antisense oligonucleotides targeting vascular endothelial growth factor. Br J Cancer,2002,87(1) : 119-126.

