Introduction
Chemokines are a class of factors or signaling proteins secreted by cells. They are called chemokines because they have the ability to induce directional movement of nearby responding cells. Existing studies have found that chemokines have four cysteine residues in their conserved positions. These residues are very important for the formation of spatial structure of chemokines. Therefore, according to the different distribution of these four cysteine residues, scientists divided the chemokines into four main subfamilies: CXC, CC, CX3C, and XC. Among them, the C chemokine protein is different from all other chemokines in that it has only two cysteine. One is at the N-terminal cysteine and the other is at the downstream half. Typical representatives of this subgroup are XCL1 (lymphokine-α) and XCL2 (lymphokine-β).
Mechanism and Function
Existing research show that the C chemokine subfamily consists of two members, XC chemokine ligand 1 (XCL1), also known as lymphokine or SCM-1 alpha, and XC chemokine ligand 2 (XCL2), also known as SCM-1 beta. Their receptor contains only one member: XCR1 (XCR1 is also known as GPR5), which is a common receptor for XCL1 and XCL2. As a chemokine receptor, XCR1 belongs to the G protein coupled receptor superfamily. This family member is characterized by the presence of seven transmembrane domains and a large number of conserved amino acids. This receptor is most closely related to the RBS11 and MIP1-alpha / RANTES receptors. It transmits signals by increasing intracellular calcium levels. Viral macrophage inflammatory protein-II is an antagonist of this receptor and blocks signaling. When the released C chemokine binds to the C chemokine receptor on the surface of the target cell, the signal begins to be transmitted across the membrane. The intracellular signal transduction of the XCR1 receptor also depends on the neighboring G protein, so when GDP binds to the receptor G protein, the G protein receptor is in an inhibited state. Binding of chemokine C to the XCR1 receptor will cause GTP molecules to replace the bound GDP and release G protein subunits. Free G protein subunits cause subsequent activation of cell membrane-associated phospholipase C (PLC), which in turn cleaves phosphatidylinositol (4,5) -diphosphate (PIP2) into inositol triphosphate (IP3) and diacylglycerol (DAG). Among them, IP3 can cause changes in intracellular calcium ions to stimulate subsequent signal transduction, and DAG can activate protein kinase C (PKC). These events can promote subsequent cascades of related intracellular signals, eventually causing the cells to move toward specific direction.
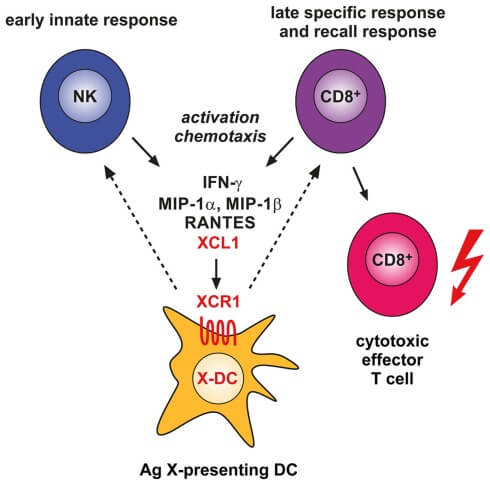 Figure 1. Involvement of the XCL1–XCR1 communication axis in the innate and adaptive cytotoxic responses to cross-presented microbial and tumor antigens.
Figure 1. Involvement of the XCL1–XCR1 communication axis in the innate and adaptive cytotoxic responses to cross-presented microbial and tumor antigens.
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- Qualitative and quantitative analysis of individual cytokines
- Qualitative and quantitative analysis of various cytokines
- Qualitative and quantitative detection of cytokines in a variety of species
- Identification and quantitative determination of cell factor antibody
Sample requirements
- Sample Types - Blood, serum, plasma, cell culture supernatant, cell lysate, cell culture medium, tissue homogenate, urine, tumor, etc.
- Sample Volume - It is optimal for at least 200µl of each sample. This volume allows for triplicate testing of each sample.
Our advantages:
- Various test methods can be selected according to samples and requirements.
- High-quality antibodies are used for detection to ensure the specificity and accuracy of the detection.
- Repeat test to ensure reproducibility and accuracy.
- Professional and efficient feedback results.
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow
For more information on chemokines C subfamily detection service or to require additional testing requirements, please contact us.
References:
- Kroczek RA, Henn V. The role of XCR1 and its ligand XCL1 in antigen cross-presentation by murine and human dendritic cells. Front. Immunol. 2012, doi: 10.3389/fifimmu.2012.00014
- Alexandre YO, Ghilas S, et al. XCR1+ dendritic cells promote memory CD8+ T cell recall upon secondary infections with Listeria monocytogenes or certain viruses. The Journal of Experimental Medicine. 2016, 213 (1): 75–92.

