Introduction
Epidermal growth factor (EGF) is an important cell growth factor secreted by the human body. It is abundant in human epithelial cells. Its functions include stimulating the growth of epithelial cells and endothelial cells, promoting the proliferation of epithelial cells and fibroblasts. In addition to regulating cell proliferation and differentiation, EGF, as a lung regulatory peptide, has anti-oxidant effects, and can regulate the function of airway epithelial cells through autocrine or paracrine methods. EGF can up-regulate the expression of γ-glutamyl transpeptidase, accelerate the transport of exogenous glutathione (GSH) into cells, promote the synthesis of GSH, improve the antioxidant capacity of cells, and eliminate reactive oxygen species. In addition, EGF has a significant regulatory effect on alveolar cell proliferation, and thus participates in the lung injury repair process. Moreover, EGF also has a certain regulatory effect on the reproductive system. EGF affects testicular development and spermatogenesis, regulates ovarian development, and promotes zygotic development. It can also be used as an embryonic nutrient factor to regulate early embryo development.
Mechanism and Function
EGF is the earliest identified growth factor with a molecular weight of 6045Da, composed of 53 amino acid residues, and contains 3 intra-chain disulfide bonds. EGF does not contain alanine, phenylalanine and lysine, and arginine at position 53 and leucine at position 52 are often hydrolyzed by protease, so more than 40 amino acids at the amino terminus of EGF have all the activity of this factor. The spatial structure of EGF molecules can be divided into relatively independent N- and C-segments, which are connected by disulfide bonds, so the spatial position of the two parts is relatively variable. Functionally, the N domain is mainly involved in receptor binding, while the C domain is mainly responsible for promoting cell growth. When EGF binds to its receptor, it can trigger a series of intracellular changes, start the mediated signaling pathway, and participate in the physiological regulation of cells.
Epidermal growth factor receptor (EGFR) is a receptor tyrosine protein kinase, and the receptor tyrosine kinase-Ras-MAPK cascade pathway is the most important pathway for epidermal growth factor-stimulated signals to be transmitted to the nucleus. After EGF binds to the receptor, it can dimerism the receptor, thereby changing the conformation of the receptor. EGFR is enhanced the tyrosine kinase activity, and phosphorylated the tyrosine residue. The receptor forms a site that can bind to the SH2 domain-containing Grb2 protein, allowing Grb2 to bind to the receptor. There are two SH3 domains in Grb2, which can bind to guanine nucleotide release factor called SOS and change its activity. Then, SOS further activates Ras, and the activated Ras acts on the MAPK signaling pathway and activates ERK. Finally, ERK transfers into the nucleus, resulting in changes in the activity of certain transcription factors, which changes the expression status of genes and the process of cell proliferation and differentiation.
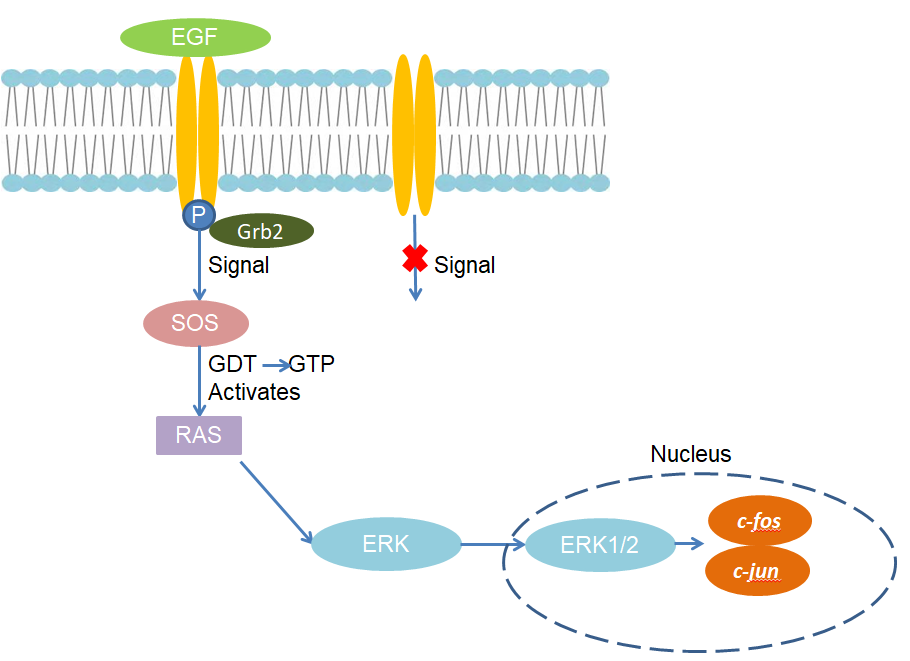 Fig 1. Mechanism of Signaling
Fig 1. Mechanism of Signaling
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- One or more cytokines cytokines qualitative and quantitative detection
- Cytokines qualitative and quantitative detection of various species
- Cytokine antibodies qualitative and quantitative detection
Sample requirements
- Sample Types-Blood, serum, plasma, cell culture supernatant, cell lysate, cell culture medium, tissue homogenate, urine, tumor, etc.
- Sample Volume - It is optimal for 50 samples. This volume allows for triplicate testing of each sample.
Our advantages:
- Different detection methods can be selected based on different samples and requirements.
- Ensure the specificity and accuracy of the test by using high quality antibodies.
- Repeat the test to ensure the repeatability and accuracy of the experimental results.
- Feedback results are accurate and efficient
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow
For more information about the EGF detection service or need other detection requirements, please contact us.

