Introduction
Platelet derived growth factor (PDGF) is a basic protein stored in platelet alpha particles. PDGF can stimulate various cells such as fibroblasts, glial cells, and smooth muscle cells that are stagnated in the G0 / G1 phase to enter the division and proliferation cycle. PDGF is a peptide regulating factor found in 1974 to stimulate the growth of tissue cells such as connective tissue. It exists in the alpha particles of platelets under normal physiological conditions. It is released and activated by disintegrating platelets when blood coagulates, and has a stimulus Specific cell chemotaxis and biological activity to promote specific cell growth. In addition, when tissues are damaged, macrophages, vascular smooth muscle cells, fibroblasts, endothelial cells, embryonic stem cells, which can also synthesize and release PDGF. When the liver is damaged, macrophages, platelets, infiltrating inflammatory cells, damaged endothelial cells, and activated hepatic stellate cells can all secrete PDGF. PDGF is a thermostable glycoprotein with a molecular weight of 30KD, and is a dimer composed of two polypeptide chains A and B connected by disulfide bonds.
Mechanism and Function
The common platelet-derived factor PDGF is a homo- or hetero-dimer formed by connecting two polypeptide chains through disulfide bonds, which makes PDGF have a variety of dimer structures, namely PDGF-AA, PDGF-BB, PDGF -AB, PDGF-CC and PDGF-DD. The molecular weight of PDGF-A chain is 16KD, and the PDGF-B chain is 14KD. A and B chain genes are located on chromosomes 7 and 22, respectively. Generally known as PDGFI, PDGF-AA, with a molecular weight of 31KD, containing 7% sugar, PDGFⅡ, PDGF-BB, with a molecular weight of 28KD, containing 4% sugar, both of which are composed of two highly homologous A and B chains. Monocytes / macrophages are cells that primarily synthesize PDGF.
PDGF must bind to the corresponding receptor on the cell membrane to exert its biological effects. PDGF receptor is composed of two subunits α and β, and its molecular weight is 170-180KD. The binding between the two and PDGF is very different. The α unit has higher affinity for the PDGFa chain and the B chain, while the β subunit has only high affinity for the B chain. Therefore, the α subunit can bind to PDGF-AA, PDGF-AB, and PDGF-BB, and the β subunit can only bind to PDGF-BB and PDGF-AB. After PDGF binds to the receptor, PDGFR containing tyrosine kinase activity is dimerized and its tyrosine residue is autophosphorylated. The activated PDGFR binds to Grb2 to activate Ras, and then Raf-1, MEK, and ERK are successively activation. Nuclear transfer of activated ERK can induce phosphorylation of multiple transcription factors and regulate cell proliferation.
The biological activity of PDGF mainly includes 4 aspects. First, it has chemotactic activity. PDGF induces migration of macrophages and fibroblasts, and has chemotaxis to neutrophils, smooth muscle cells, and fibroblasts. In the early stages of trauma, it can promote the accumulation of surrounding cells to the wound site, cooperate with platelet coagulation, activate the immune system at the site of the wound, and lay the foundation for wound repair. Second, it has vasoconstrictive activity. PDGF can cause vasoconstriction and is a stronger vasoactive substance than angiotensin II. In the early stage of trauma, it can stimulate the capillary of the wound site to contract rapidly, reduce the blood pressure and flow rate of the wound site, promote blood coagulation, and create conditions for wound repair. At the same time, PDGF can induce the division and proliferation of damaged epithelial cells and endothelial cells, promote the formation and regeneration of blood vessels, and provide a guarantee for wound repair. Third, it has the mitogenic effect. PDGF can stimulate the proliferation and proliferation of vascular smooth muscle cells, fibroblasts and glial cells. PDGF transmits cell signals by activating PDGF receptor transmembrane proteins, and stimulates various cells such as fibroblasts, glial cells, and smooth muscle cells that have stalled in the G0 / G1 phase to enter the division and proliferation cycle. Fourth, it is involved in phosphatase activation and prostaglandin metabolism. When PDGF interacts with cells with receptors, it can induce the phosphoinositide cycle and the release of arachidonic acid, and promote the production of prostaglandins, PGI2 and PGE2. Increased PGI2 and PGE2 may accelerate bone resorption and increase their vasodilatory and antiplatelet activity.
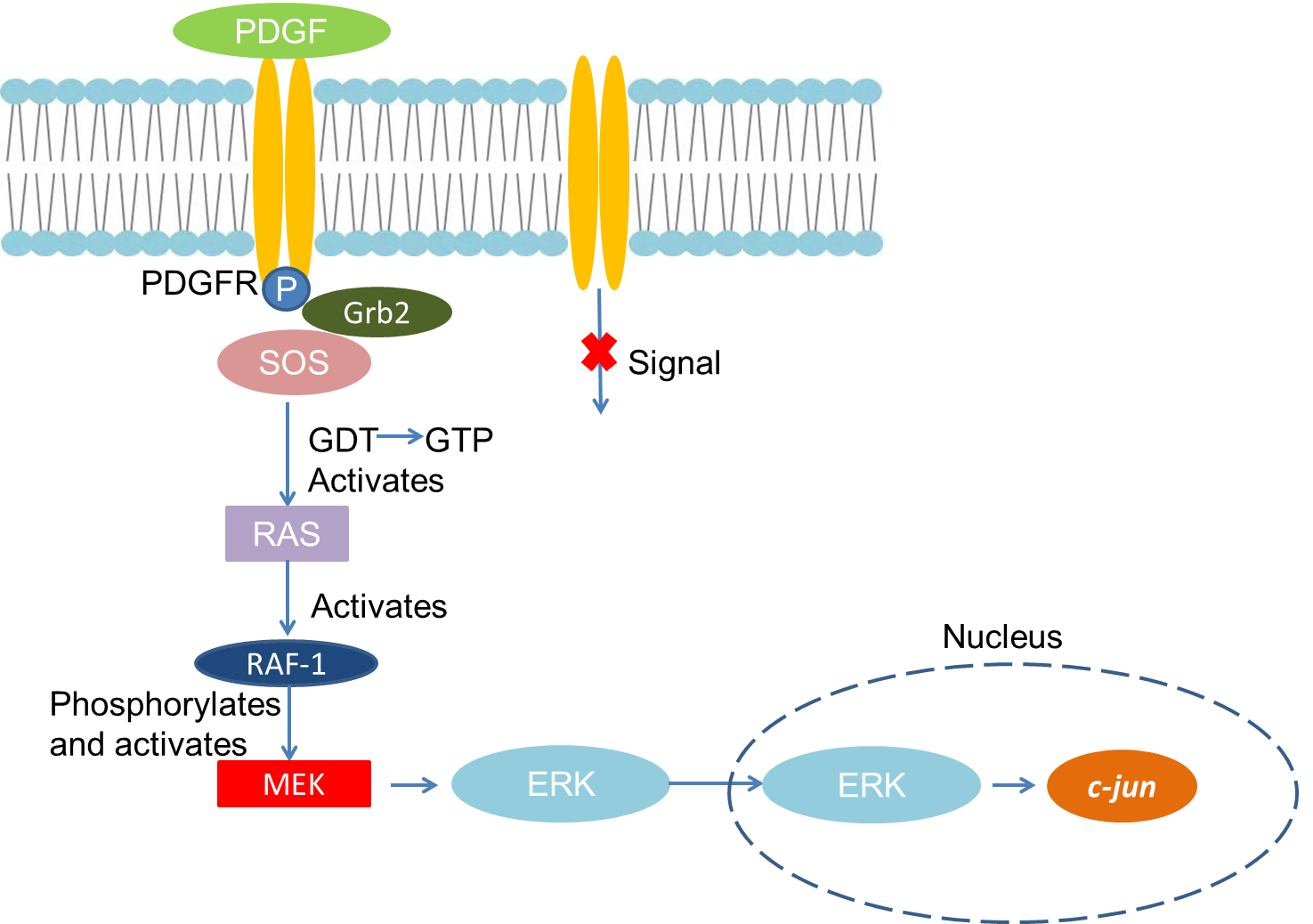 Fig 1. Mechanism of Signaling
Fig 1. Mechanism of Signaling
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- One or more cytokines cytokines qualitative and quantitative detection
- Cytokines qualitative and quantitative detection of various species
- Cytokine antibodies qualitative and quantitative detection
Sample requirements
- Sample Types-Blood, serum, plasma, cell culture supernatant, cell lysate, cell culture medium, tissue homogenate, urine, tumor, etc.
- Sample Volume - It is optimal for 50 samples. This volume allows for triplicate testing of each sample.
Our advantages:
- Different detection methods can be selected based on different samples and requirements.
- Ensure the specificity and accuracy of the test by using high quality antibodies.
- Repeat the test to ensure the repeatability and accuracy of the experimental results.
- Feedback results are accurate and efficient
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow

For more information about the PDGF detection service or need other detection requirements, please contact us.
References:
- Heidaran MA, et al. A delection in the extracellular domain of the α platelet-derived growth factor differently impairs PDGF-AA and PDGF-BB binding affinities. J Biol Chem, 1996, 267:2884.
- Graves L M, et al. A antagonizes platelet-derived growth factor-induced signaling by mitogen-activated protein kinase in human arterial smooth muscle cells. Proc Natl Acad Sci USA, 1993, 90:10300.

