Introduction
Granulocyte colony-stimulating factor (G-CSF) is a glycoprotein containing 174 amino acids and a molecular weight of about 20,000. It is mainly produced by monocytes and macrophages activated endotoxin, TNF-α and IFN-γ. The G-CSF gene is 2.5 kb in length, including 5 exons and 4 introns. G-CSF has 5 cysteine, and Cys 36 and Cys42, Cys74 and Cys64 form two disulfide bonds. Cys17 is an unpaired cysteine, and disulfide bonds are essential for maintaining the biological function of G-CSF. Human and mouse G-CSF have 73% homology at the amino acid level and have biological activities that cross each other. G-CSF mainly acts on the proliferation, differentiation, and activation of neutrophil hematopoietic cells. Recombinant human granulocyte macrophage colony-stimulating factor (rhGM-CSF) acts on hematopoietic progenitor cells to promote their proliferation and differentiation. Its important role is to stimulate the maturation of granulocytes and monocytes, promote the release of mature cells to peripheral blood, and promote multiple functions of macrophages and phagocytic cells. G-CSF is mainly used for the prevention and treatment of leukopenia caused by tumor radiotherapy or chemotherapy, the treatment of bone marrow hematopoietic dysfunction and myelodysplastic syndrome, and the prevention of potential infection complications of leukopenia.
Mechanism and Function
Granulocyte colony-stimulating factor (G-CSF) has been discovered, purified, and named for more than 20 years. It was named G-CSF in 1983, which is also currently called colony-stimulating factor 3 (CSF-3). In addition to hematopoietic tissue, G-CSF can also be produced by vascular endothelial cells, fibroblasts, nerve cells. Malignant solid tumors such as bladder, gastrointestinal tumors and glioma cells can also secrete a large amount of G-CSF, shorten the proliferation and differentiation cycle of hematopoietic stem cells, increase the level of neutrophils in circulating blood, and promote cell survival and migration.
G-CSFR is a transmembrane protein, including an extracellular immunoglobulin-like (Ig) domain, a cytokine receptor homeodomain (CRH), and three fibronectin type III (FNIII) modules. The intracellular domain consists of It is composed of 3 amino acid sequences (Box1-3) and is the key structure of signal transduction. Among them, Box3 located at the far end of the domain contains 4 tyrosine residue phosphorylation sites, which are Y704, Y729, Y744, and Y764, respectively. Phosphorylation sites can recruit many signaling molecules containing Src homeodomain 2 (SH2) or phosphotyrosine binding (PTB) domains to play a specific role in division and differentiation.
After G-CSF binds to G-CSFR, two adjacent receptor-ligand dimers form a tetramer complex, which activates JAK1, JAK2, and Tyk2 in the JAKs kinase family in the cell. Intracellular domain undergoes tyrosine phosphorylation and further recruits SH2 domain-containing signaling proteins such as STATs, cytokine signal inhibitor 3 (SOCS3), and growth factor receptor binding protein 2 (Grb2), which affect the expression of core genes or mitochondrial changes, thereby altering the processes of cell differentiation, proliferation, apoptosis, migration or inflammation. Among them, the STATs family includes STAT1, STAT3, and STAT5 participate in G-CSF-induced cell proliferation or differentiation. STAT3 binds to the tyrosine site at the distal end of the receptor, and has different activation processes under physiological and pathological conditions. Cyclin-dependent kinase (CDK) inhibitor p27 Kip1 plays a role in promoting cell differentiation by binding to STAT3. STAT1 and STAT5 bind and act on the intracellular proximal domain of the receptor. STAT5 reaches its peak and rapidly after G-CSFR activation. Decay for about 30 min. The SOCS protein is a negative regulator of the JAKs / STATs pathway. The SH2 domain of SOCS3 binds to Y729, which is phosphorylated at the distal end of the receptor membrane, and inhibits JAKs enzyme activity to reduce the level of STATs phosphorylation. It plays an important role in the process of proliferation and differentiation.
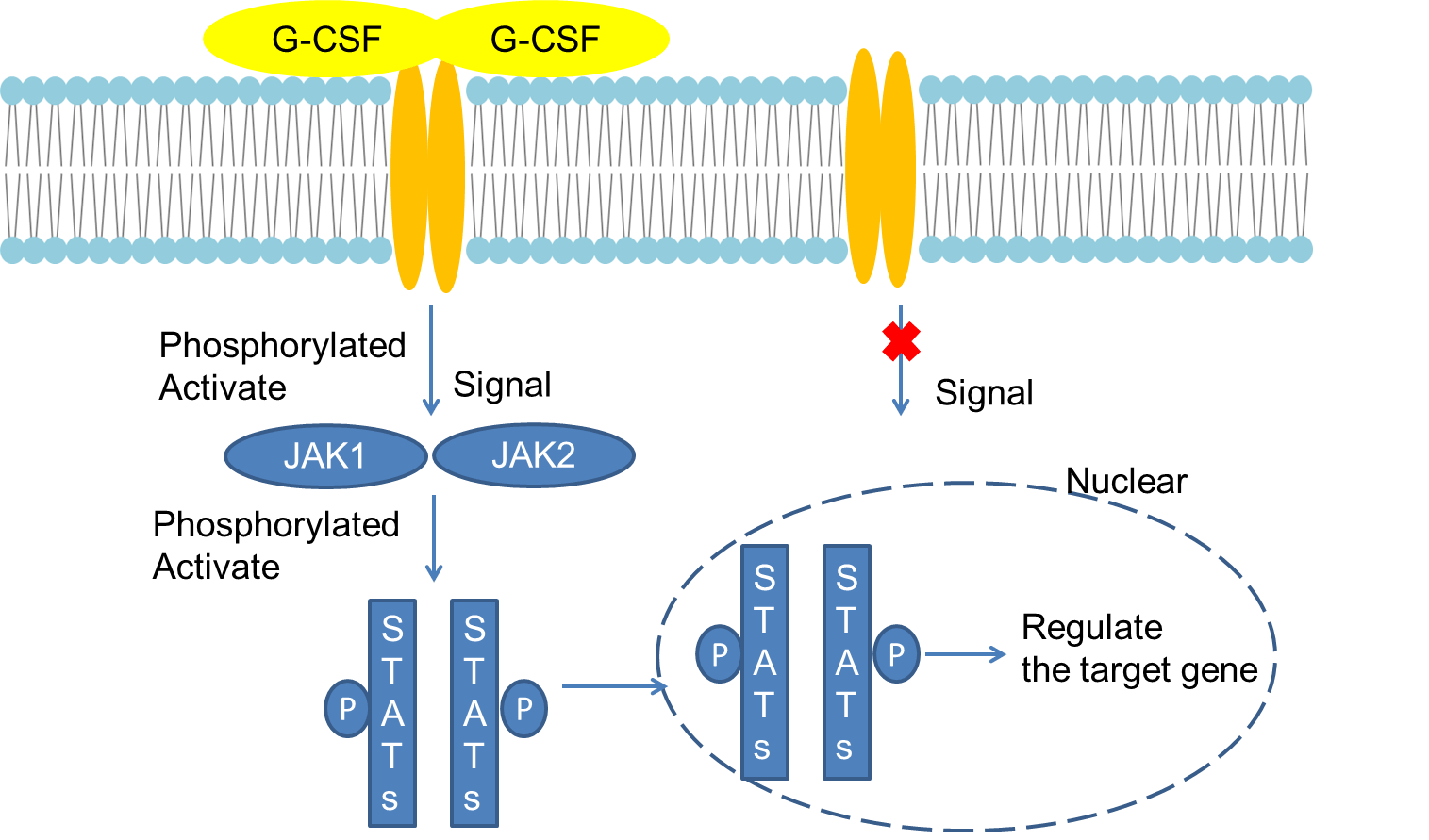 Fig 1. Mechanism of Signaling
Fig 1. Mechanism of Signaling
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- One or more cytokines cytokines qualitative and quantitative detection
- Cytokines qualitative and quantitative detection of various species
- Cytokine antibodies qualitative and quantitative detection
Sample requirements
- Sample Types- Serum, plasma and related fluid samples, etc.
- Sample Volume - It is optimal for 50 samples. This volume allows for triplicate testing of each sample.
Our advantages:
- Different detection methods can be selected based on different samples and requirements.
- Ensure the specificity and accuracy of the test by using high quality antibodies.
- Repeat the test to ensure the repeatability and accuracy of the experimental results.
- Feedback results are accurate and efficient
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow
For more information about the G-CSF detection service or need other detection requirements, please contact us.
References:
- DWIVEDI P,GREIS KD. Granulocyte Colony Stimulating Factor Receptor(G-CSFR)signaling in severe congenital neutropenia,chronic neutrophilic leukemia and related malignancies[J]. Exp Hematol,2017.DOI:10.1016/j.exphem.2016.10.008.
- CHATZIMPALOGLOU A, KOLOSOV M, ECKOLS TK, et al. Synthetic and biological studies of phaeosphaerides [J]. J Org Chem, 2014, 79(9): 4043-4054.

