Introduction
Chemokines, also known as chemokines or chemical hormones, are small cytokines or signaling proteins secreted by cells. Their name comes from their ability to induce directional chemotaxis of nearby responding cells. The chemokine family has more than 40 members. Based on the position of two conserved cysteines in the N-terminal region, chemokines can be divided into CXC, C, CC, and CX3C subfamilies. CXC chemokine subfamily is called CXC because its two cysteine at the N-terminal is separated by amino acids. Seventeen different CXC chemokines have been described in mammals. These CXC chemokines can be divided into two classes. Among them, the chemokine with a specific amino acid sequence of glutamic acid-leucine-arginine (ELR) immediately before the first cysteine of CXC is a type (ELR Positive) and those without ELR are another type (ELR Negative). Previous studies have found that ELR-positive CXC chemokines can interact with chemokine receptors CXCR1 and CXCR2 and specifically induce neutrophil migration. ELX-negative CXC chemokines tend to attract lymphocytes.
Mechanism and Function
The main role of chemokines is to induce directional movement of cells. Cells attracted by chemokines follow chemokine concentration toward the source of chemokine. In the process of immune surveillance, different chemokines control different types of immune cells. These are called homeostasis chemokines and are produced and secreted without the need to stimulate the source cells. CXC chemokines secreted by cells need to bind to chemokine receptors on the cell surface to achieve inducible functions. The CXC chemokine receptor is a seven-transmembrane G protein-coupled receptor that specifically binds and responds to cytokines of the CXC chemokine family. Seven CXC chemokine receptors are currently known in mammals, from CXCR1 to CXCR7.
The signals generated by the binding of CXC chemokines to receptors need to be transduced with the help of G proteins near the intracellular receptors. Generally, G proteins exist as heterotrimers, which are composed of three different subunits: Gα, Gβ, and Gγ. At this time, the GDP molecule is bound to the G protein subunit, and the G protein is in an inactive state. When the receptor is activated, G protein is recruited to the intracellular receptor, thereby replaced GDP by GTP molecules and dissociating different G protein subunits. The Gβ subunit activates a phospholipase C (PLC) on the cell membrane, which can cleave phosphatidylinositol (4,5) -bisphosphate (PIP2) to form two second messenger molecules, called inositol Triphosphate (IP3) and diacylglycerol (DAG); DAG activates another enzyme called protein kinase C (PKC), and IP3 triggers the release of calcium from the endoplasmic reticulum. These events promote many signal cascades and affect cellular responses.
At the same time, the protein subunit Gα directly activates phosphotyrosine kinase (PTK). Activated PTK, on the one hand, activates the MAPK pathway, and on the other hand, it also phosphorylates serine and threonine residues of the chemokine receptor, causing its desensitization or inactivation. The activated MAPK pathway activates specific cellular mechanisms, including chemotaxis, degranulation, release of superoxide anions, and changes in the activity of cell adhesion molecules (called integrins). CXC chemokines can also activate small Rho GTPase signaling pathways, which mainly include these including the most widely studied Rho GTPases, RhoA, Rac1, 2 and Cdc42, and their complexes with effector and regulatory factors. They act as conformational switches in various signal transduction pathways that regulate a variety of cellular functions, participate in downstream effector interactions that control cytoskeleton rearrangement, regulate nucleotide exchange, the hydrolysis cycle, and the distribution between cell membrane and cytoplasm. In addition, the combination of CXC and CXCR also activates NF-κB, Camp / PKA, and other pathways, which together with the aforementioned pathways regulate cell adhesion, polarization, and directional movement.
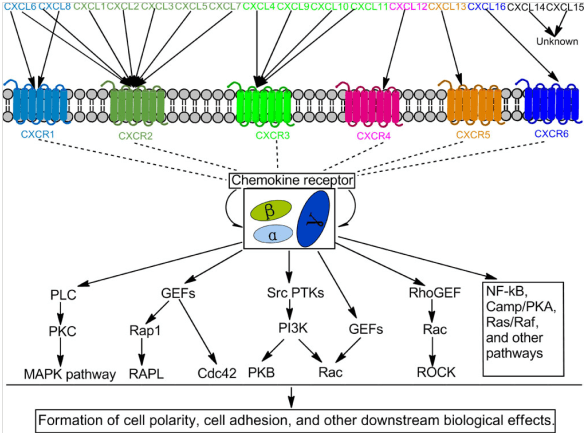 Figure 1. Multiple signaling pathways induced by CXC and CXCR.
Figure 1. Multiple signaling pathways induced by CXC and CXCR.
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- Single cytokines be analyzed at qualitative and quantitative
- Multiple cytokines be analyzed at qualitative and quantitative
- Qualitative and quantitative test of cytokines in various species
- Qualitative and quantitative test of cytokine antibodies
Sample requirements
- Sample Types - Blood, serum, plasma, cell culture supernatant, cell lysate, cell culture medium, tissue homogenate, urine, tumor, etc.
- Sample Volume - It is optimal for at least 200 µl of each sample. This volume allows for triplicate testing of each sample.
Our advantages:
- Multiple test methods can be selected based on different samples and requirements.
- High-quality antibodies are used in the test process to improve the specificity and accuracy of the detection.
- Repeat detection to ensure reproducibility and accuracy of test results.
- Professional and efficient feedback results.
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow
For more information on chemokines CXC subfamily detection service or to require additional testing requirements, please contact us.
References:
- Qingchao Zhu. et al. The role of CXC chemokines and their receptors in the progression and treatment of tumors. J Mol Hist. 2012, DOI 10.1007/s10735-012-9435-x.
- Albert Zlotnik, et al. Chemokines: A New Classification Review System and Their Role in Immunity. Immunity. 2000, 12, 121–127.
- H Nomiyama, et al. Organization of the chemokine genes in the human and mouse major clusters of CC and CXC chemokines: diversification between the two species. Genes and Immunity. 2001, 2, 110–113.

