Introduction
Interferon-alpha (IFN-α) is a group of structurally similar, functionally similar low molecular glycoproteins produced by antiviral response when the immune cells are infected with the virus. There are many IFN-α subtypes with molecular weights ranging from 15 to 21 kDa. IFN-α mainly have three functions: broad-spectrum antiviral effect, immunomodulatory effect, and anti-tumor effect, but the relative biological activity may be different. Since IFN-α has various anti-proliferative and anti-tumor activities, it is one of the most commonly used cytokine treatments for cancer patients with hairy cell leukemia, kidney cancer, Kaposi's sarcoma and other malignant tumors. Under physiological conditions, low levels of IFN-a can be detected. However, in infections and various pathological conditions, its production is significantly enhanced, making IFN-α a major disease marker.
Mechanism and Function
Interferon-α (IFN-α), a pro-inflammatory cytokine, currently has at least 23 genes found clustered in the 9p22 region of human chromosomes with a total length of 1-2 kb. The gene structure consists of a 5' non-coding region, a secretion signal peptide, an interferon polypeptide, and a 3' non-coding region. About 60% of the IFN-α region constitutes five α-helices, and further forms a globular protein with two disulfide bonds, which is important for the correct folding and biological activity of IFN-α.
The IFN-α gene is usually induced to be expressed, that is, the IFN-α gene is not expressed under normal conditions. IFN-α gene is induced when stimulated by stimulating factors including viruses, lipopolysaccharides, polypeptides, and dsRNA. IFN-α binds to the IFN-α receptor on the effector cell membrane and transduces the signal into the cell, resulting in the activation of tyrosine protein kinase in the cytoplasm, which in turn causes three free IFN-stimulated gene factors (ISGF-3α) subunits phosphorylation in the cytoplasm. The phosphorylated three ISGF-3α subunits bind to each other to form a triple complex that enters the nucleus along with the ISGF-3γ subunit. ISGF-3γ binds to the interferon-stimulated response element (ISRE), and the ISGF-3α subunit activates ISRE, allowing effector cells to produce antiviral protein (AVP). AVPs are some species-specific enzymes, mainly including 2', 5'-oligoadenylate synthetase, protein kinase, and phosphodiesterase. 2', 5'-oligoadenylate synthetase polymerizes ATP into oligoadenylate due to dsRNA, which activates the potential nuclease F in the cell, degrades the mRNA of the virus, and selectively degrades the mRNA of the virus. Protein kinase phosphorylates eIF-2, which prevents the initiation of the synthesis of the polypeptide chain of the viral protein. Phosphodiesterase removes the CCA at the end of the tRNA and blocks protein translation of the virus. Besides, IFN-α enhances HLA-I antigen expression on infected hepatocyte membranes and regulates IL-1, IL-2, and tumor necrosis factor (TNF), while also activates NK cells, K cells, and macrophages to exert their biological effects.
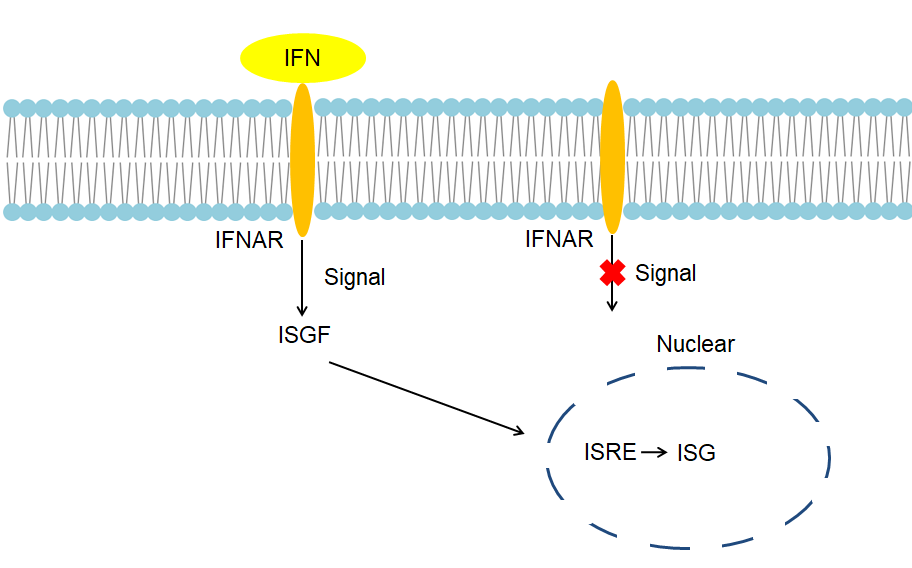 Fig 1. Mechanism of Signaling
Fig 1. Mechanism of Signaling
Creative Proteomics can provide cytokine detection platform for scientific research. According to different purposes, our dedicated analysts will customize exclusive solutions for you. We aim to provide customers with high-quality and convenient services to help you accelerate the progress of your project.
Our cytokine detection service includes but is not limited to:
- One or more cytokines cytokines qualitative and quantitative detection
- Cytokines qualitative and quantitative detection of various species
- Cytokine antibodies qualitative and quantitative detection
Sample requirements
- Serum, buffered solution or cell culture medium, etc.
- Sample Volume - It is optimal for 5µl of each sample. And 500 assay point kit allows to run 500 wells in 96-well or 384-well format.
Our advantages:
- Different detection methods can be selected based on different samples and requirements.
- Ensure the specificity and accuracy of the test by using high quality antibodies.
- Repeat the test to ensure the repeatability and accuracy of the experimental results.
- Feedback results are accurate and efficient
Technology platform:
We mainly provide the Luminex cytokine detection platform. Luminex uses fluorescently encoded microspheres with specific antibodies to different target molecules. The different microspheres can be combined freely to a certain extent so that up to 100 analytes can be tested multiple times simultaneously in a single experiment.
The Luminex cytokine assay platform has the following advantages:
- Multiple detection: simultaneous detection of 100 biological targets
- Short experiment time: 1-3 weeks
- High sensitivity: the lower limit of accurate quantification is as low as 0.1 pg/mL
- Save samples: only need a sample volume as low as 25 μL
- Time saving: the experiment process only takes 4 hours
For your different needs, we can also provide the following detection methods:
- Enzyme-linked immunosorbent assay (ELISA)
- Flow cytometry
Workflow
 Fig 2. workflow
Fig 2. workflow
For more information about the IFN-α detection service or need other detection requirements, please contact us.
References:
- Murayama, G.,et al., Enhanced IFN-alpha production is associated with increased TLR7 retention in the lysosomes of palasmacytoid dendritic cells in systemic lupus erythematosus. Arthritis Res Ther, 2017. 19(1): p. 234.
- Fitzgibbon, M., et al., Endocannabinoid modulation of inflammatory hyperalgesia in the IFN-alpha mouse model of depression. Brain Behav Immun, 2019. 82: p. 372-381.

