Cytokines are generally small in molecular weight and high in biological activity and are mainly produced by immune or non-immune cells (e.g., vascular endothelial cells, epidermal cells, and fibroblasts) upon stimulation. Cytokines can interact with each other to form networks that are involved in immune response, inflammatory response and cell proliferation and growth.
Multiple cytokines form a network associated with disease susceptibility. When immune tolerance is disrupted, cytokine homeostasis is lost and the local target tissue environment shifts to a pro-inflammatory state and leads to tissue damage, resulting in systemic immune diseases. Uncovering the relationship between cytokines and diseases at the cellular and molecular levels, especially the study of the pathogenesis of autoimmune diseases and tumors, can help provide a basis for guiding clinical treatment and diagnosis.
- Cytokines in Cytokine Release Syndrome (CRS)
CRS is a systemic inflammatory response that can be triggered by a variety of factors, such as infectious, autoimmune inflammatory, and medical causes. Cytokines of CRS can be classified as inflammatory effectors, inflammatory initiators, inflammatory modulators, chemokines and colony-stimulating factors.
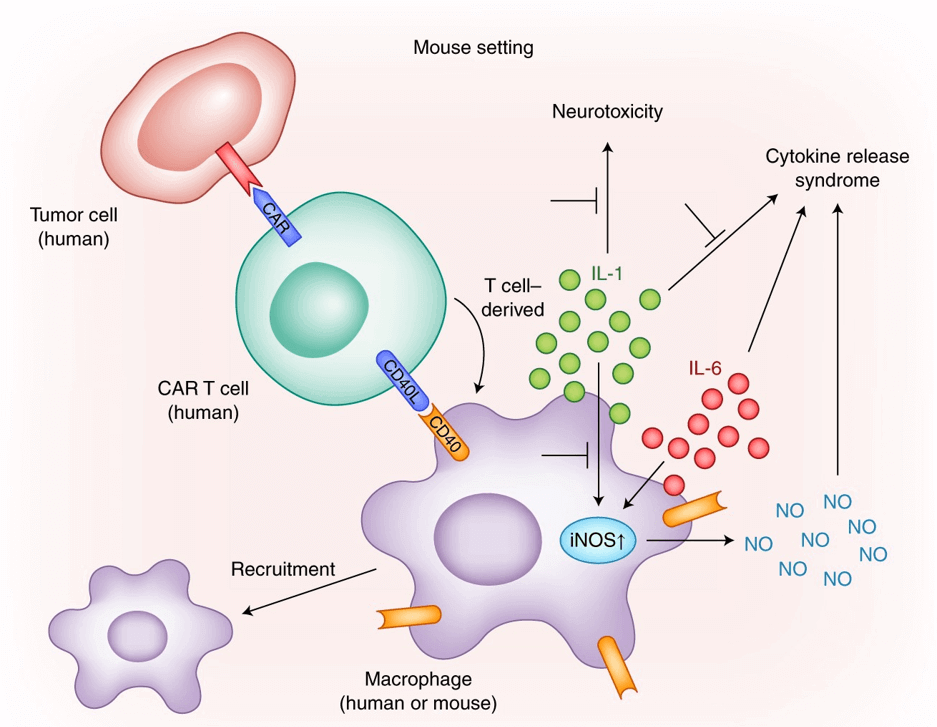 Modeling CRS after CAR T cell transfer in mice (Rooney et al., 2018)
Modeling CRS after CAR T cell transfer in mice (Rooney et al., 2018)
CRS inflammatory effectors are cytokines that cause major clinical symptoms. For example, INF-γ, which causes fever and hematopoiesis, TNF-α, which causes fever and depression, and I-6, which causes fever and acute kidney injury and NK cell insufficiency.
CRS inflammation initiation factor refers to the recognition by macrophage pattern after the virus enters the body, and the receptor recognition causes the assembly of inflammasome, which releases mature IL-1B and IL-18 cytokines and initiates inflammation.
Inflammatory regulators mainly refer to the mutual regulation and balancing effect of pro-inflammatory factors such as IL-2 and anti-inflammatory factors such as IL-10 and IL-4 as the disease progresses.
CRS colony-stimulating factor is produced by activated T cells and macrophages, which stimulate granulocyte and macrophage colony formation. Granulocyte colony-stimulating factor (G-CSF) stimulates granulocyte function, as well as IL-7, enhances cytotoxic activity of macrophages, and induces cytokine secretion by monocytes.
The above factors hierarchically trigger and promote the CRS process.
RA is a systemic immune-related polyarticular inflammatory disease that destroys joints and causes physical disability, and involves T cells, B cells, lymphocytes, macrophages, and various cytokines such as IL-1B, IL-6, IL-12, IL-15, and transforming growth factor (TGF-3). In addition to producing autoantibodies and forming immune complexes, B cells promote disease progression by producing cytokines (e.g., IL-6, IL-10, etc.) and chemokines. B-cell-derived cytokines regulate follicular DC and lymphatic activation, providing a regulatory feedback loop for T-cell-macrophage and T-cell-B-cell interactions. Therefore, T-cell and B-cell signaling pathways and pathway-related cytokines are hot spots for drug approval and development in RA arthritis.
- Cytokines in Asthma
Asthma is one of the most common chronic diseases in children and adults. Asthma pathology is characterized by narrowing of the airways and chronic inflammation of the airway bark with plasma extravasation, edema and influx of inflammatory cells such as eosinophils, neutrophils, lymphocytes, macrophages and mast cells.
T cells develop into follicular helper T cells (Tfh) in the lung draining lymph nodes, which secrete IL-4 to B cells to control the synthesis of the immunoglobulin IgE. IgE binds to receptors and triggers an inflammatory response, which is an important pathological mechanism in asthma attacks. IL-4 and IL-13 associated with tissue eosinophilia, and L-13 associated with bronchial hyperresponsiveness, which exacerbate the progression of asthma. The expression of key adhesion molecules such as vascular cell adhesion molecule-1 (VCAM-1) or intercellular adhesion molecule 1 (ICAM-1) contributes to eosinophil extravasation and exacerbates the pathological process of asthma.
- Cytokines in Inflammatory Bowel Disease (IBD)
IBD is a chronic inflammatory disease of the gastrointestinal tract, including Crohn's disease (CD) and ulcerative colitis (UC). Gastrointestinal homeostasis is maintained by the interaction of mononuclear phagocytes (MNP), Foxp3+ Treg cells, B cells, and cytokines from intestinal epithelialcells (IECs) and immune cells. Once imbalanced, IBD can be induced.
STAT3-induced cytokines, such as IL-22, IL-6, and IL-17, promote IEC cell survival and anti-microbial defense. Microbial and metabolite-induced IL-10, TGF-β1 play a role in tolerance through MNP-Treg cell interactions. IL-1B, IL-18 and TNF-α produced by MNP and IECs are key pro-inflammatory factors at the onset of the colitis process. IL-23-driven CD4+ T cell response is a key pathological feature of IBD. The characteristic T cell population of IL-23 is involved in driving intestinal inflammation through the production of the effector cytokines IFN-γ, IL-17 and GM-CSF and further stimulates the myeloid response in the local intestine.
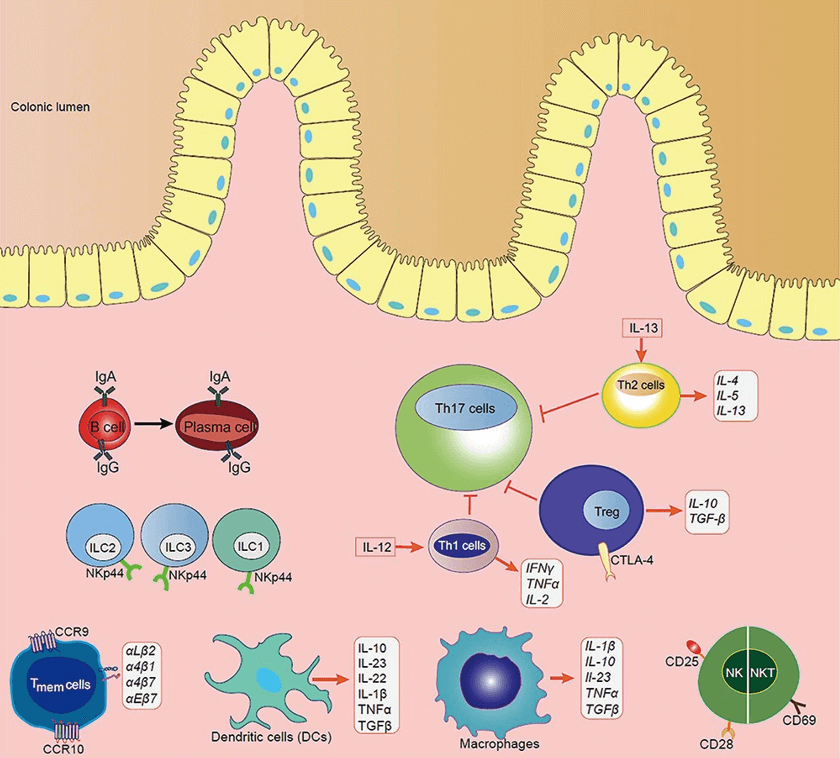 The disturbance of the immune cell on the progression of IBD (Lu et al., 2022).
The disturbance of the immune cell on the progression of IBD (Lu et al., 2022).
- Cytokines in Chronic Obstructive Pulmonary Disease (COPD)
COPD is characterized by incomplete reversible airflow limitation and is primarily driven by a type 2 immune response, including increased airway eosinophils, increased secretion of Th2 cells and type II innate lymphoid cells (ILC2s), and IL-4, IL-5, and IL-13. TNF-α concentrations in sputum and blood were increased in patients with acute exacerbation COPD. The expression of IL-1β and IL-32 correlated with the severity of the disease.
Under normal physiological conditions, cytokines in body fluids or tissues regulate the development, differentiation and function of immune cells at very low levels, and regulate the immune response of the body, playing an important role in the maintenance of body homeostasis.
The balance of anti-inflammatory and inflammatory factors is disrupted when the organism is abnormal. For example, when a storm of inflammatory factors occurs, a variety of pro-inflammatory cytokines are produced rapidly and in large quantities in body fluids, the corresponding cellular and intracellular signaling pathways are changed, and the disease process is affected and changed.
Inflammatory factors are not only involved in the development of inflammatory immune diseases, but also play a role in cardiovascular diseases, metabolic diseases and CNS diseases.
Cytokine Assay
Inflammation assays are important for basic disease research and for understanding the process of disease development.
Luminex xMAP technology allows the detection of multiple biomarkers using a single sample, or multiple substances simultaneously in a single experiment.
With a range of 3 - 4 orders of magnitude, Luminex protein measurement has the advantage of being highly specific, rapid, and reproducible for the simultaneous detection of multiple cytokines or proteins.
References:
- Rooney, C., & Sauer, T. (2018). Modeling cytokine release syndrome. Nature Medicine, 24(6), 705-706.
- Lu, Q., Yang, M. F., et al. (2022). Immunology of Inflammatory Bowel Disease: Molecular Mechanisms and Therapeutics. Journal of Inflammation Research, 15, 1825.

