Who this guide is for
- Human cohort studies selecting a multiplex cardiovascular biomarker assay with consistent cross-batch performance
- Preclinical teams using mouse models and needing a scalable cardiovascular biomarker panel strategy
- CRO/biotech programs outsourcing to a cardiovascular disease biomarker assay provider: cardiovascular disease biomarker assay
Cohort constraints that shape multiplex cardiovascular biomarker assay selection
Timeline and batch effects
- Long enrollment windows and plate-to-plate drift control
- Lot continuity strategy and change-control checkpoints
- QC anchor samples repeated across runs
Serum/plasma matrix reality
- Serum vs plasma comparability planning across sites
- Anticoagulant selection and analyte compatibility
- Interference triage: hemolysis, lipemia, icterus
Sensitivity, dynamic range, and missingness
- Low-abundance cytokines driving "below LLOQ" gaps
- High-abundance markers driving "above ULOQ" saturation
- Split-panel vs dilution-rule decisions
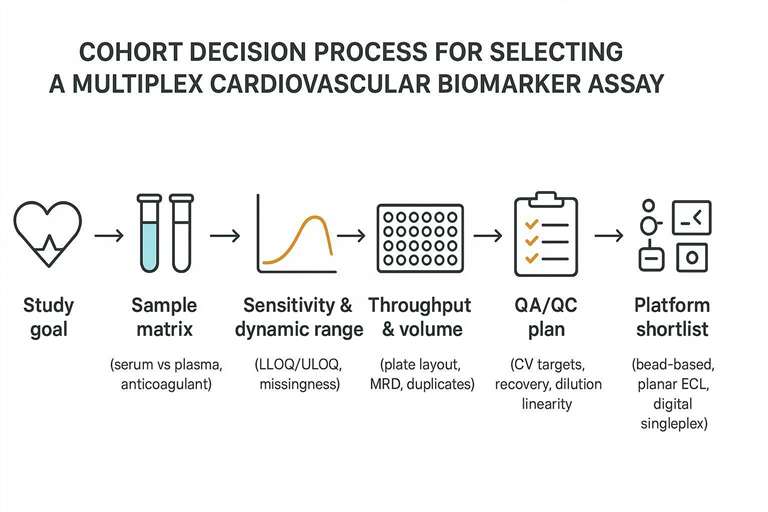 Cohort-first decision path from study goal to platform shortlist and QA/QC plan.
Cohort-first decision path from study goal to platform shortlist and QA/QC plan.
Define the goal before comparing platforms
Endpoint pressure
- Primary endpoint vs exploratory marker stacking
- Longitudinal change detection vs cross-sectional stratification
- Missingness tolerance for event-driven analyses
Operational throughput
- Sample volume budgeting across repeats and panels
- Plate design logic: calibrators, blanks, QCs, selective duplicates
- Deliverables for downstream stats: concentrations + QC flags + curve metrics
Shortlist assay formats without turning this into a generic "vs" page
Multiplex vs singleplex (cohort trade-off)
- Multiplex for breadth, volume efficiency, and high N
- Singleplex for endpoint-critical ultra-low analytes
- Hybrid strategy: multiplex core + targeted high-sensitivity add-ons
Outsourcing-ready service pathways on cytokine.creative-proteomics.com
- Custom multiplex execution via Luminex cytokine detection service
- Flexible panel scoping via cytokine panel service
- Cohort-ready project entry point via cardiovascular disease biomarker assay
Human vs mouse cohort translation (plan panels by species)
- Human clinical cohort panel options via human cardiovascular disease panel service
- Mouse model panel options via mouse cardiovascular disease panel service
Platform comparison table (cohort selection lens)
| Cohort decision lens | Bead-based multiplex (Luminex-style) | Planar multiplex (ECL-style) | Digital immunoassay | Singleplex ELISA |
|---|---|---|---|---|
| Best fit | Broad panels, limited volume, scalable throughput | Mid-size panels with strong low-end performance | Ultra-low endpoint analytes | Few targets, deep validation |
| Cohort scaling | High | High | Medium | Medium–Low |
| Serum/plasma risk | Matrix effects vary by analyte | Panel availability constraints | Throughput constraints | Volume/time burden |
| Typical deployment | Core multiplex cardiovascular biomarker panel | Complementary multiplex panel | Add-on for 1–3 endpoints | Validation subset |
 Cohort-centric platform comparison across throughput, volume, sensitivity, and matrix risk.
Cohort-centric platform comparison across throughput, volume, sensitivity, and matrix risk.
Serum/plasma checkpoints that protect cohort biomarker data
Serum vs plasma choice rules
- Site collection feasibility and SOP locking
- Storage stability over long durations
- Anticoagulant harmonization across enrollment sites
Pre-analytical gates
- Time-to-spin window alignment
- Aliquot strategy to reduce freeze–thaw cycles
- Handling reference for serum/plasma via serum & plasma cytokine assay
Sample handling checklist table (cohort standardization)
| Step | Minimum standard | Failure mode | Mitigation |
|---|---|---|---|
| Collection | Same tube type per site | Mixed anticoagulants | Lock site SOP |
| Processing | Fixed time window | Activation/proteolysis | Time-stamp workflow |
| Aliquoting | Multiple small aliquots | Excess freeze–thaw | Aliquot plan by panel count |
| Storage | Stable long-term temp | Excursions | Monitored freezers |
| Shipping | Validated cold chain | Partial thaw | Dry ice + logger policy |
 A practical checklist to reduce pre-analytical variability and batch artifacts.
A practical checklist to reduce pre-analytical variability and batch artifacts.
QA/QC plan that makes multiplex cardiovascular biomarker assays "cohort-usable"
Define acceptance criteria before the first plate
- LLOQ/ULOQ coverage targets by analyte group
- Precision targets: intra-/inter-assay CV%
- Matrix checks: spike recovery + dilution linearity
Recommended RUO targets grounded in bioanalytical guidance: intra-assay CV typically ≤20–25%; inter-assay CV ≤20–30%; spike recovery ~80–120%; evaluate dilution linearity/parallelism on incurred samples with acceptable fit to the calibration model. For conceptual anchors, see the U.S. FDA's 2018 guidance in Bioanalytical Method Validation Guidance for Industry and the EMA/ICH M10 bioanalytical method validation guideline.
Quantification thresholds and reporting language
- Locked rules for values < LLOQ and > ULOQ
- Curve acceptance and rerun triggers
- Threshold framing aligned with LLD/LLOQ/ULOQ in Luminex cytokine assays
Examples (fit-for-purpose, RUO):
- <LLOQ: report as "<LLOQ" with the numeric LLOQ; do not impute centrally unless prespecified.
- ULOQ: apply validated dilution or report as ">ULOQ" with ULOQ value when re-measurement is infeasible.
- Curve acceptance: require back-calculated calibrators within ±20% at LLOQ/ULOQ and ±15% elsewhere, with run-level QC pass rules as per guidance.
Controls that scale across sites and timepoints
- QC anchors repeated across plates/runs
- Bridge controls for lot changes and site waves
- Plate map discipline for comparability
Operationalizing drift control in cohorts:
- Select 2–3 pooled serum/plasma anchors spanning low/mid/high concentrations; place on every plate for the entire study.
- When lots change or new sites/waves begin, include 20–30 previously characterized samples as bridging controls and evaluate agreement using Deming regression and Bland–Altman plots; consider correction factors only when acceptance windows (e.g., slope 0.8–1.25; bias within clinically/biologically acceptable bands) are not met.
- Randomize plate positions; bracket unknowns with QCs and verify absence of edge and carryover effects.
Decision rules table (shortlist in minutes)
| If your cohort looks like… | Prioritize | Deprioritize | Next step |
|---|---|---|---|
| Many endpoints + limited volume + many timepoints | Multiplex core panel + dilution rules | Singleplex-only expansion | Start with human cardiovascular disease panel service |
| Mouse model discovery feeding a human cohort | Species-matched panels + shared pathway logic | One-size-fits-all markers | Use mouse cardiovascular disease panel service |
| Primary endpoint is ultra-low abundance | Endpoint-first assay + multiplex secondary | One panel for everything | Lock endpoint, then panelize |
| Multi-site + long storage + staggered shipping | Bridging controls + drift rules + QC anchors | Minimal QC | Add stability and bridging plan |
What to ask an outsource partner before committing
Feasibility and matrix-fit questions
- Serum/plasma validation evidence per analyte
- Interference handling and exception rules
- Range planning and dilution strategy confirmation
QC transparency and deliverables
- Standard curve exports and QC summary per run
- Raw signal availability and data dictionary
- Missingness rationale: < LLOQ vs assay failure vs sample QC
Related cluster links (non-overlapping intent)
- Panel composition logic: How to Design a Cardiovascular Biomarker Panel
- Sample logistics deep dive: Serum/Plasma for Cardiovascular Biomarker Assays: Shipping Checklist
- QC acceptance criteria deep dive: Multiplex Biomarker Assay QA/QC Acceptance Criteria
FAQ
What is the best multiplex cardiovascular biomarker assay for cohort studies?
The "best" is the platform and panel that meet your sensitivity and dynamic-range needs, throughput targets, and predefined QA/QC acceptance criteria for your serum/plasma matrix. Use RUO fit‑for‑purpose validation aligned to FDA/ICH guidance to confirm suitability.
Should I use serum or plasma for a multiplex cardiovascular biomarker cohort study?
Choose the matrix that matches site feasibility and long‑term stability, then lock anticoagulant and processing windows across sites and validate matrix‑specific performance and HIL (hemolysis/lipemia/icterus) risks.
How many biomarkers can I include in one multiplex cardiovascular biomarker panel?
It depends on analyte ranges, matrix effects, and dilution strategy; confirm via feasibility with cohort‑like samples and consider split‑panels when ULOQ saturation and LLOQ missingness co‑exist.
Which QA/QC metrics matter most for cohort comparability?
LLOQ coverage, inter‑assay CV%, spike recovery, and dilution linearity/parallelism are critical because they drive missingness patterns and cross‑batch drift detection.
How do I reduce batch effects in long-running cohort biomarker programs?
Use QC anchor samples on every plate, add bridging controls for lot/site/timepoint transitions, and enforce locked curve/QC acceptance criteria with rerun rules; assess agreement with Deming regression and Bland–Altman analysis.
Can I align mouse model biomarker panels to human cohort readouts?
Yes—start with species‑matched panels and shared pathway logic; validate cross‑species assay performance and document interpretation limits before scaling to cohorts.
What causes high missingness (values below LLOQ) in cytokine panels, and how can I prevent it?
True low abundance and matrix suppression are common causes; mitigate by optimizing MRD, splitting panels to avoid range conflicts, improving pre‑analytics, and verifying LLOQ suitability during feasibility.
Should I split my panel into two panels to improve dynamic range and completeness?
Yes when you observe simultaneous high‑end saturation and low‑end missingness; use a multiplex core for breadth and add targeted singleplex/digital assays for ultra‑low endpoints.
References:
- Günther, A., et al. 2020. Comparison of bead-based fluorescence versus planar electrochemiluminescence multiplex immunoassays for measuring cytokines in human plasma. Frontiers in Immunology 11:572634. https://doi.org/10.3389/fimmu.2020.572634
- Liu, M. Y., et al. 2005. Multiplexed Analysis of Biomarkers Related to Obesity and the Metabolic Syndrome in Human Plasma, Using the Luminex-100 System. Clinical Chemistry 51(7):1102–1109. https://doi.org/10.1373/clinchem.2004.047084
- U.S. Food and Drug Administration. 2018. Bioanalytical Method Validation Guidance for Industry. https://www.fda.gov/files/drugs/published/Bioanalytical-Method-Validation-Guidance-for-Industry.pdf
- European Medicines Agency (ICH). ICH guideline M10 on bioanalytical method validation and study sample analysis (Step 5). https://www.ema.europa.eu/en/documents/scientific-guideline/ich-guideline-m10-bioanalytical-method-validation-step-5_en.pdf

