Panel design goals for cardiovascular biomarker studies
Designing a cardiovascular biomarker panel is ultimately about decision support. Start with a clear biological question and translate it into measurable readouts. Use an endpoints-first logic (risk, progression, response, safety) to define which markers are non‑negotiable and which are optional. Finally, insist on cohort-ready consistency—plan for batch effects, missingness, and repeatability from day one—so your cardiovascular biomarker panel design scales from pilot to validation.
- Clear biological question → measurable biomarker readouts
- Endpoints-first panel logic (risk, progression, response, safety)
- Cohort-ready consistency (batch effects, missingness, repeatability)
Start with a pathway map: inflammation + adhesion + oxidative stress
A panel that mixes complementary pathways is more interpretable and more resilient across diverse cohorts. Think in lanes: inflammation (innate activation), endothelial activation/adhesion (vascular interface), and oxidative stress (redox imbalance and plaque biology). Below is a compact guide to each.
Inflammation biomarkers (innate immune activation)
- Acute-phase signal vs cytokine network: hs‑CRP provides a robust, acute‑phase context, while cytokines (e.g., IL‑6, TNF family) capture upstream signaling that often sits closer to mechanism and treatment response.
- Cytokines tied to vascular inflammation and risk stratification: The IL‑6 axis features prominently in cardiovascular inflammation biology and has been linked to adverse outcomes in research settings, reinforcing its role in panel design for hypothesis‑driven work (see Ridker & Rane, 2021 in References).
Adhesion molecule biomarkers (endothelial activation)
- Leukocyte trafficking and vascular injury signals: sICAM‑1 and sVCAM‑1 reflect endothelial activation and leukocyte adhesion. Selectins (E‑ and P‑selectin) add rolling/platelet–endothelium context.
- Soluble adhesion molecules for systemic endothelial activation context: Individually, effect sizes may be modest; in a multi‑pathway panel they help distinguish vascular interface status and complement inflammatory and redox readouts (see Blankenberg et al., 2001; Pickett et al., 2023).
Oxidative stress biomarkers (redox imbalance and plaque biology)
- Oxidant enzymes and lipid/protein oxidation proxies: Myeloperoxidase (MPO) bridges inflammation and oxidative stress and is frequently associated with adverse cardiovascular events in acute settings; oxidation adducts and AGEs can serve as proxies of redox imbalance.
- Oxidative stress markers aligned to atherosclerosis and remodeling themes: Treat oxidative markers as complementary signals that contextualize endothelial and inflammatory axes, especially when studying plaque biology and remodeling.
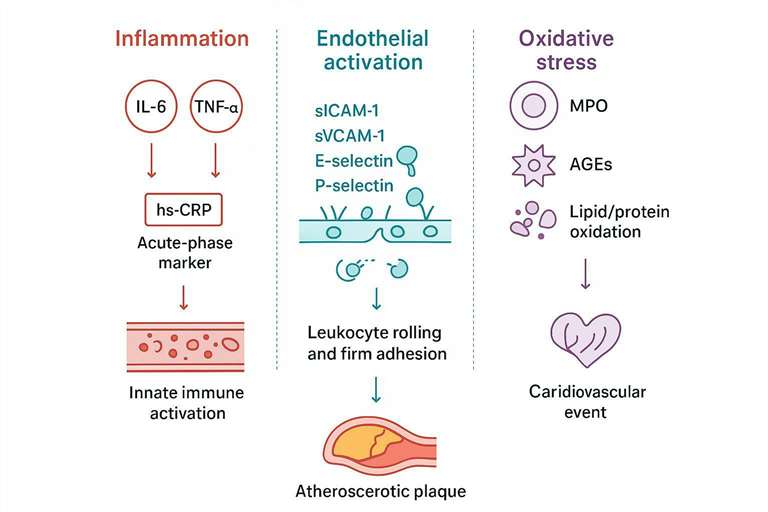 A pathway-first blueprint for designing a cardiovascular biomarker panel.
A pathway-first blueprint for designing a cardiovascular biomarker panel.
Define the panel "job": what decisions the data must support
Hypothesis-driven vs exploratory panel design
- Must-have markers for primary hypotheses: Reserve "Core" slots for markers tied directly to risk/progression endpoints (e.g., hs‑CRP; sICAM‑1/sVCAM‑1; possibly MPO depending on cohort).
- Optional markers for mechanism expansion: Add "Mechanism" candidates (e.g., IL‑1/IL‑6 axis, selectins, oxidation proxies) to deepen interpretation.
- Split panels to protect data completeness: If expected concentrations and dilution rules diverge widely, split into two runs to minimize out‑of‑range values and re‑runs.
Sample and matrix reality (serum/plasma)
- Sample volume budget per subject and timepoint: Lock the per‑timepoint volume before finalizing plex size; remember replication and potential reruns.
- Matrix effects planning and feasibility gating: Validate the chosen matrix for interference and stability; define rejection rules for hemolysis/lipemia.
- Logistics alignment via serum & plasma cytokine assay: This resource outlines practical serum/plasma handling considerations you can adapt to cardiovascular panels.
Throughput and scaling needs
- Pilot vs validation vs full cohort scaling: Start small to confirm range coverage and missingness, then scale with the same controls and bridge standards.
- Plate layout discipline: Include calibrators, QCs (high/low), and bridge controls on every plate; randomize samples to mitigate plate position effects.
- Deliverables: Provide concentrations with QC flags and assay metadata (curve parameters, lot IDs, plate maps, rerun notes) to support downstream statistics.
Choose a panel strategy: custom vs preconfigured
When a custom cardiovascular biomarker panel is the right fit
- Mixed pathways (inflammation + adhesion + oxidative stress) that don't cleanly match a catalog set.
- Tight endpoint mapping and subgroup analyses where you need to tune analyte ranges and volumes.
- Feasibility scoping via cytokine panel service: Use a feasibility pilot to test dilution rules, LLOQ coverage, and matrix effects before locking the final build.
When a disease-focused preconfigured panel fits better
- Standard cardiovascular readouts and faster start with known marker sets.
- Cross-study comparability when harmonizing with existing cohorts.
- Human cohort option via human cardiovascular disease panel service.
- Mouse model option via mouse cardiovascular disease panel service.
Build the marker list with a decision framework (avoid keyword cannibalization)
"Core / Context / Mechanism" marker tiers
- Core: Endpoint‑linked markers (risk/progression/response) that must meet range and precision goals in your matrix.
- Context: Confounder‑aware markers reflecting systemic inflammation and endothelial status that stabilize interpretation.
- Mechanism: Pathway probes that illuminate biology and can explain heterogeneity in Core readouts.
Range compatibility and missingness control
- Low‑abundance cytokines vs high‑abundance proteins: Separate by dilution rules to minimize OOR and preserve precision.
- Dilution rules and split‑panel triggers: Pre‑define dilution steps (e.g., 1:2, 1:4, 1:10) and thresholds that trigger a separate run.
- LLOQ/ULOQ planning aligned with LLD/LLOQ/ULOQ in Luminex cytokine assays: Align panel composition to expected concentration ranges to limit missingness.
Assay format fit: multiplex panel vs singleplex add-ons
- Multiplex cytokine/biomarker panels maximize breadth and volume efficiency, ideal for an inflammation biomarkers cardiovascular panel anchored in risk/progression.
- Singleplex add‑ons suit endpoint‑critical, ultra‑low analytes (e.g., IL‑6 in general-population cohorts) where ultrasensitivity is required.
- Hybrid design tied to cohort selection logic; for platform background, see Luminex cytokine detection service.
Example panels table (Inflammation + Adhesion + Oxidative Stress)
| Panel type | Best for | Inflammation module | Adhesion module | Oxidative stress module |
|---|---|---|---|---|
| Core panel | Risk/progression screening | CRP + key cytokines | sICAM-1 / sVCAM-1 | MPO or AGEs |
| Mechanism panel | Pathway interpretation | IL-1/IL-6 axis + TNF-family | Selectins + adhesion | MPO + oxidative damage proxies |
| Response panel | Treatment/perturbation studies | Cytokine shifts + acute-phase | Endothelial activation shift | Redox shift indicators |
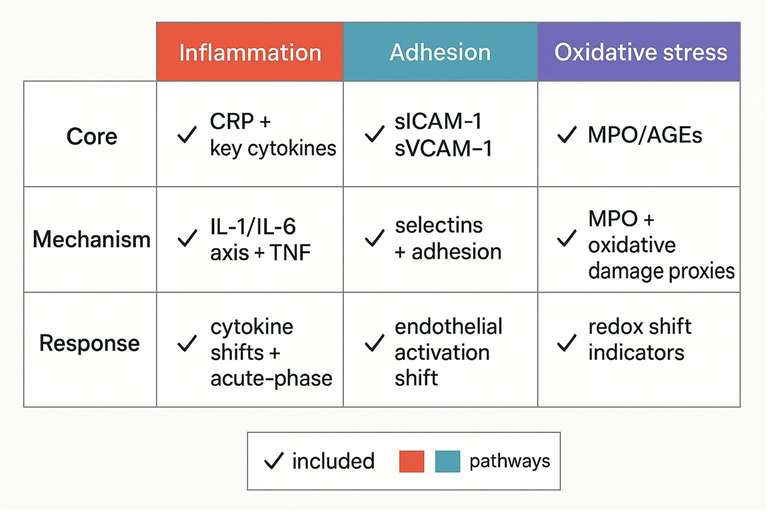 Panel templates mapped to objectives: core, mechanism, and response.
Panel templates mapped to objectives: core, mechanism, and response.
Species-specific panel design: human cohorts vs mouse models
Human cohort biomarker panel considerations
- Baseline heterogeneity and comorbidity confounding: Expect wider concentration distributions; stratify by age/sex and medication status when possible.
- Medication effects and age/sex stratification: Record cardiometabolic therapies and anti‑inflammatory agents; interpret shifts accordingly.
- Service pathway via human cardiovascular disease panel service to source species‑matched analytes and matrices.
Mouse cardiovascular biomarker panel considerations
- Strain, model, and timepoint alignment: Match sampling windows to model kinetics (e.g., diet‑induced atherosclerosis vs injury models).
- Translational mapping to human pathways: Map by pathway (inflammation, adhesion, oxidative stress) rather than forcing one‑to‑one marker identity.
- Service pathway via mouse cardiovascular disease panel service for species‑specific feasibility.
Pre-analytical and QC gates to lock panel performance
Sample handling gates that protect signal integrity
- Collection tube standardization across sites; harmonize draw timing and fasting state.
- Freeze–thaw limits and aliquoting plan: Pre‑aliquot to avoid unnecessary cycles; document any deviation.
- Shipping checklist alignment: Temperature control, transit time, and receipt logs; adapt from serum & plasma cytokine assay guidance.
QA/QC acceptance criteria for multiplex cardiovascular biomarker panels
- Precision targets (illustrative): aim for intra‑assay ≤10–20% CV and inter‑assay ≤15–25% CV, assay‑dependent and defined a priori.
- Spike recovery and dilution linearity rules: Expect recoveries around 80–120% and confirm parallelism across dilutions before full rollout.
- Curve and rerun triggers: Out‑of‑range rates, failed control recoveries, or poor duplicate CVs prompt predefined reruns and, if needed, singleplex follow‑ups.
- For cardiovascular program scoping and deliverables, see cardiovascular disease biomarker assay.
Panel feasibility checklist table (what must be confirmed before launch)
| Feasibility item | Why it matters | Output |
|---|---|---|
| Matrix fit (serum/plasma) | Prevents interference-driven artifacts | Matrix validation notes + rejection rules |
| Range coverage | Prevents missingness and saturation | Dilution plan + split-panel decisions |
| Cross-reactivity risk | Protects multiplex specificity | Compatibility map + exclusions |
| Control strategy | Protects batch comparability | Plate map + bridge/QC anchors |
| Data package | Protects downstream stats | Concentrations + QC flags + metadata |
 A study-ready workflow for cardiovascular biomarker panel design and execution.
A study-ready workflow for cardiovascular biomarker panel design and execution.
Execution pathways on cytokine.creative-proteomics.com
- Custom multiplex build-out via cytokine panel service
- Multiplex readout platform via Luminex cytokine detection service
- Full project entry point via cardiovascular disease biomarker assay
FAQ
How many biomarkers should a cardiovascular biomarker panel include?
A panel should include only as many markers as you can measure with reliable range coverage (LLOQ–ULOQ) and acceptable missingness in your chosen matrix. Prioritize Core markers tied to your endpoint, then add Context/Mechanism markers if volume and feasibility allow.
What is the difference between a cytokine panel and a cardiovascular biomarker panel?
A cytokine panel centers on inflammatory signaling. A cardiovascular biomarker panel extends to endothelial activation/adhesion (e.g., sICAM‑1, sVCAM‑1, selectins) and oxidative stress (e.g., MPO, oxidation proxies) to better reflect plaque biology and vascular interface states.
Should I design one large multiplex panel or split into two panels?
Split when analyte ranges or dilution rules diverge, matrix effects are substantial, or endpoint‑critical markers risk out‑of‑range values. Protect data completeness first; bridge controls can preserve comparability across runs.
Which sample type is better for panel-based cardiovascular biomarker assays: serum or plasma?
Either can work. Choose the matrix your cohort can standardize consistently and that demonstrates stable performance for the selected analytes in a feasibility pilot (parallelism, recovery, precision).
Can I align mouse biomarker panels with human cohort panels?
Yes. Use species‑matched assays and map by pathway (inflammation, adhesion, oxidative stress). Interpret differences through baseline physiology and model context rather than enforcing one‑to‑one markers.
What QC metrics should I require for a multiplex cardiovascular biomarker panel?
Require clear LLOQ/ULOQ coverage, predefined intra‑/inter‑assay precision targets, and matrix checks such as spike recovery and dilution linearity, with rerun rules defined up front.
References:
- Ridker, P. M., Rane, M. Interleukin‑6 Signaling and Anti‑Interleukin‑6 Therapeutics in Cardiovascular Disease. Circulation Research. 2021;128(11):1728–1746. DOI: 10.1161/CIRCRESAHA.121.319077
- Pickett, J. R., Wu, Y., Zacchi, L. F., Ta, H. T. Targeting endothelial vascular cell adhesion molecule‑1 in atherosclerosis: drug discovery and development of VCAM‑1–directed therapeutics. Cardiovascular Research. 2023;119(13):2278–2293. DOI: 10.1093/cvr/cvad130
- Günther, A., et al. Comparison of bead‑based fluorescence versus planar electrochemiluminescence multiplex immunoassays for measuring cytokines in human plasma. Frontiers in Immunology. 2020;11:572634. DOI: 10.3389/fimmu.2020.572634

