Measuring the senescence-associated secretory phenotype (SASP) sounds straightforward—until you try to compare results across cell types, inducers, timepoints, and matrices. The SASP is a moving target: a heterogeneous, dynamic mix of cytokines, chemokines, matrix remodelers, and other secreted proteins that evolves with context. If you're building a multiplex SASP panel to support mechanism studies, senolytic screening, or translational work, the right choices start with a clear question, a fit-for-purpose panel, and a disciplined interpretation framework that pairs protein readouts with orthogonal senescence markers.
This practical guide walks through what to measure, which sample type to choose, how to design and validate a multiplex SASP panel across common study scenarios, and how to interpret changes without overclaiming senolytic efficacy. We use a multi-platform perspective (Luminex xMAP, MSD, Olink, Simoa) with Luminex as a recurring workflow example, and we ground key methodological statements in peer-reviewed sources.
Why Measuring the SASP Is Not as Simple as Picking a Few Cytokines
SASP profiling is one of the most widely used functional readouts in senescence research, but it isn't a single universal signature. Reviews emphasize that SASP composition varies by cell type, senescence trigger (e.g., irradiation, oncogenic stress, chemotherapy), and time after induction, which means a panel that works beautifully in one model can mislead in another. Practical guides should therefore help you decide what to measure, in which sample type, and how to interpret any change alongside orthogonal markers.
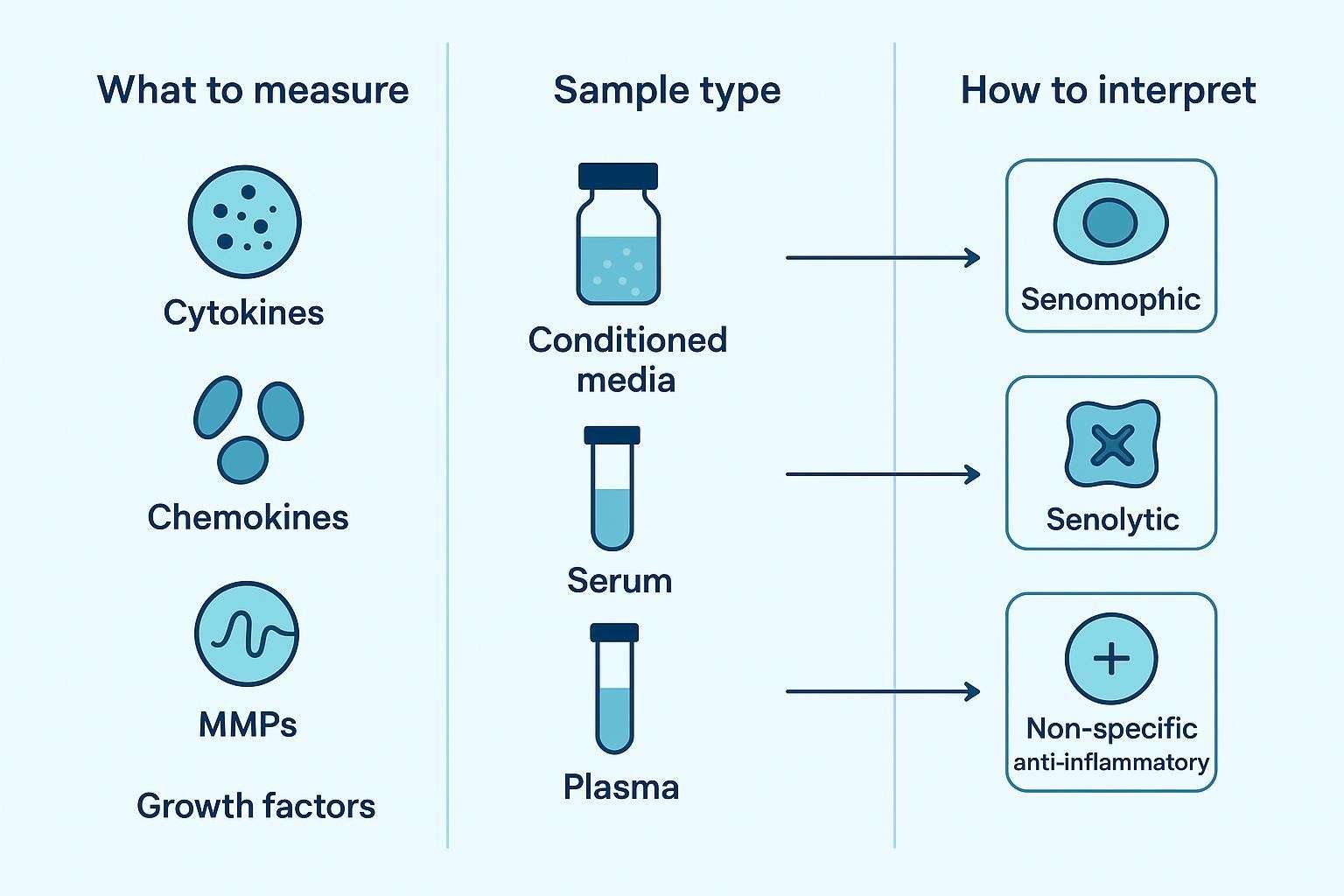
Researchers typically ask four questions at the outset: which SASP factors belong in a practical multiplex panel; whether to assay conditioned media, serum, or plasma; which readouts are most informative for senescence vs senolytic studies; and how to distinguish senomorphic activity from true senolytic effects. The sections below address each decision in turn.
What the SASP Actually Includes
The SASP encompasses more than a short list of "usual suspects." Across models, commonly measured classes include inflammatory cytokines, chemokines, matrix-remodeling proteins and inhibitors, and selected growth factors and secreted modifiers. A focused panel that blends these classes generally provides higher interpretability than a long, unfocused list.
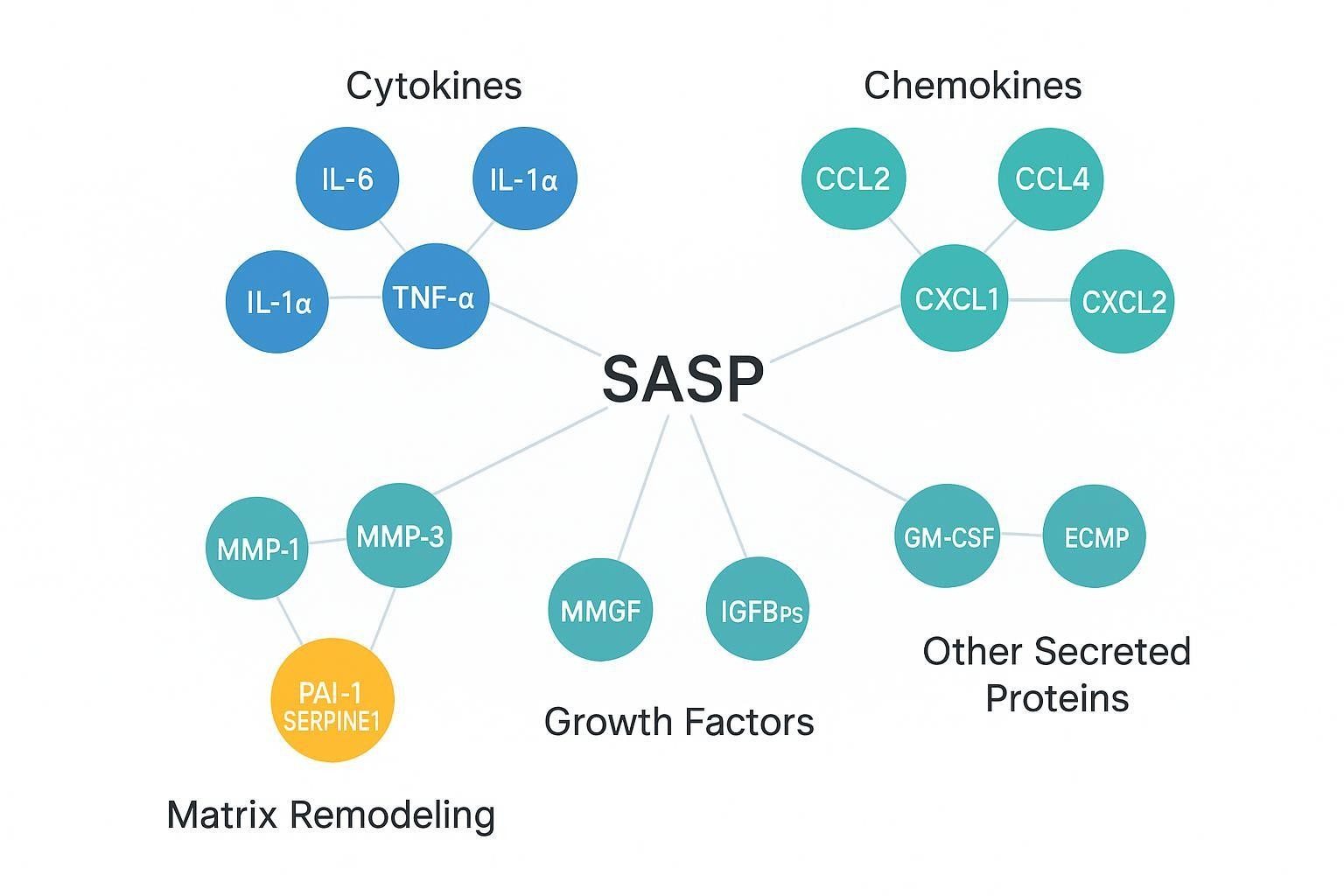
Table — Representative SASP marker classes and why they matter
| Class | Representative analytes | Why include in SASP measurement |
|---|---|---|
| Inflammatory cytokines | IL-6; IL-8/CXCL8; IL-1α; IL-1β; context-dependent TNF-α | High-frequency SASP components; often rise early; anchor the inflammatory axis |
| Chemokines | CCL2/MCP-1; CCL3; CCL4; CXCL1; CXCL2 | Capture paracrine signaling and immune-cell recruitment; strengthen panel-level coherence |
| Matrix remodeling and inhibitors | MMP-1; MMP-3; MMP-10; PAI-1/SERPINE1 | Reflect extracellular remodeling linked to senescent niches; PAI-1 is frequently associated with senescence pathways |
| Growth factors and others | GM-CSF; VEGF; IGFBPs | Broaden biological context; useful in discovery or TIS models |
A few notes on scope: IL-6/IL-8/IL-1 family members are "common but not sufficient." Chemokines help capture the paracrine nature of SASP. Matrix remodelers (MMPs) and PAI-1/SERPINE1 link secretory changes to extracellular matrix dynamics. In discovery-oriented studies, growth factors and IGFBPs may reveal pathway-specific modulation, but breadth should be balanced with detectability and reproducibility.
Which SASP Markers Are Most Useful for a Multiplex SASP Panel
A focused core panel for routine SASP measurement generally blends high-yield inflammatory cytokines (e.g., IL-6, IL-8/CXCL8, IL-1α/β), chemokines such as CCL2/MCP-1 and CXCL1/2, and a limited set of matrix remodelers (e.g., MMP-1, MMP-3, and PAI-1/SERPINE1). That mix tends to be detectable across common in vitro models and is informative for both induction confirmation and intervention studies. Expanding beyond the core is warranted when you're in a discovery phase, comparing multiple senescence inducers, or interrogating fibrosis, tumor microenvironment, or "inflammaging" biology.
Marker choice should be driven by the study objective. For induction confirmation, prioritize robust, early-rising cytokines and chemokines. For longitudinal tracking, ensure components remain quantifiable across expected ranges and timepoints. For senomorphic studies, emphasize pattern-level dampening across the panel rather than one-marker drops. For senolytic response evaluation, include factors expected to fall when senescent-cell burden is reduced—and plan to corroborate with cell-state markers and viability-aware controls.
Which Sample Type to Use: Conditioned Media, Serum, or Plasma
Sample type changes both biological meaning and analytical behavior. Conditioned media (CM) records direct secretory output from senescent cells in vitro and is ideal for mechanistic comparisons across untreated, induced, and intervention-treated conditions. Blood-derived matrices are more relevant for in vivo or translational questions but integrate many biological sources and pre-analytical influences; interpretation must therefore be conservative.
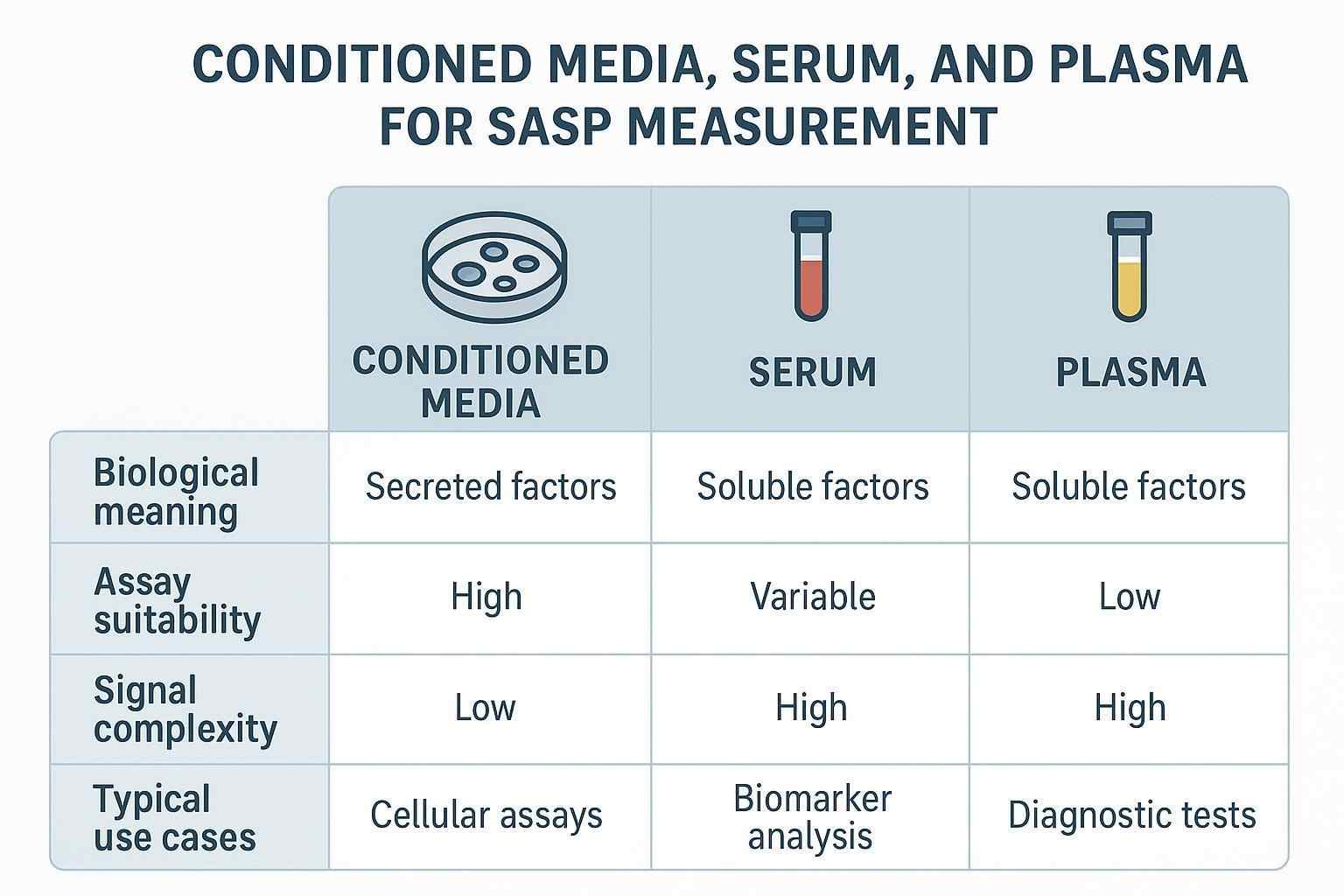
Table — Sample-type considerations for SASP studies
| Aspect | Conditioned media | Serum | Plasma |
|---|---|---|---|
| Biological meaning | Direct cell-autonomous secretion | Systemic mixture; includes clot-release factors | Systemic mixture; anticoagulated to limit clotting |
| Assay suitability | Clean matrix but watch media background | Strong signals for some cytokines; platelet activation inflates some markers | Better for platelet-sensitive targets with appropriate tubes (e.g., CTAD) |
| Signal complexity | Lower; controlled by cell number and media | Higher; influenced by clotting and background inflammation | Higher; influenced by tube/additives and processing |
| Typical use cases | In vitro induction and mechanism studies | Translational cohorts; clinical-adjacent contexts (cautious interpretation) | Translational cohorts; preferred for platelet-sensitive/protease-sensitive targets |
Two practical caveats. First, serum and plasma are not interchangeable for protein measurements. Serum clotting releases platelet cargos (e.g., sCD40L, PDGF family), which can inflate apparent levels of certain factors; CTAD plasma or rigorously prepared platelet-poor plasma can mitigate such artifacts in platelet-sensitive analytes. Second, processing delays and temperature control influence measured cytokines; document tube type, time-to-spin, centrifugation settings, and freeze–thaw cycles, and include stability checks in method validation.
How to Design a Multiplex SASP Panel for Different Study Scenarios
Design begins with the experimental question and the matrix. Below are scenario-focused principles that translate across platforms.
Panels for in vitro senescence induction studies should use a focused set of high-yield inflammatory and remodeling markers, aligned to the cell type and induction method, and paired with clear untreated vs induced controls. Panels for senolytic screening should emphasize repeatable markers that support compound ranking across runs and include factors that would reasonably decrease if senescent-cell burden falls. Senomorphic studies benefit from designs that highlight suppression of secretory activity without assuming cell elimination; look for coherent pattern dampening rather than a single cytokine drop. For translational or circulating-biomarker studies, use a conservative interpretation framework and favor a backbone of detectable, well-behaved markers with documented pre-analytical controls.
As a concrete, neutral example, teams often operationalize an in vitro or small-animal multiplex SASP panel on Luminex xMAP to balance breadth, sample volume, and throughput, then confirm key observations on an orthogonal platform or with orthogonal cellular assays. For readers who need a refresher on the technology fundamentals, see What is Luminex xMAP Technology? . When configuring or verifying a focused SASP set, a service built around xMAP can conserve limited sample volume while supporting multi-species workstreams (serum/plasma, conditioned media, tissue lysates) (Knowledge Base Source). If you're looking for a worked configuration example to align a small, fit-for-purpose panel with your model and species, the Luminex Cytokine Panel Service page outlines typical design options (Knowledge Base Source).
Choosing platforms: practical comparison for SASP work
Each immunoassay platform has strengths and trade-offs. A balanced comparison helps match platform to objective.
Table — Platform characteristics for multiplex SASP measurement (evidence-tied, qualitative)
| Platform | Detection principle & strengths | Typical trade-offs for SASP use |
|---|---|---|
| Luminex xMAP | Bead-based fluorescence multiplexing; broad plex capacity; efficient sample use; mature ecosystem for cytokines | Sensitivity and bias are analyte- and matrix-dependent; cross-reactivity control is critical; validation per analyte required |
| MSD (electrochemiluminescence) | Planar ECL with strong dynamic range and often favorable LLOQs in head-to-heads for some analytes; good precision reported | Lower plex than bead-based; reported concentration bias vs bead assays in matched samples; kit availability by species varies |
| Olink (PEA) | Dual-antibody proximity extension with NGS/qPCR readout; scalable, discovery-friendly panels and built-in normalization | Indirect units for discovery panels; orthogonal confirmation typically required; direct numeric cross-platform equivalence is non-trivial |
| Simoa | Digital ultrasensitive single-molecule counting; excellent for very low-abundance targets | Lower plex; higher per-assay complexity; best reserved for a few critical low-level markers |
For a deeper dive on xMAP fundamentals that inform assay setup and data handling, see What is Luminex xMAP Technology?. When executing a multiplex run and documenting QC (e.g., spike–recovery, dilution linearity, and precision), practical setup notes and sample-volume guidance are summarized in the Luminex Cytokine Detection Service overview (Knowledge Base Source).
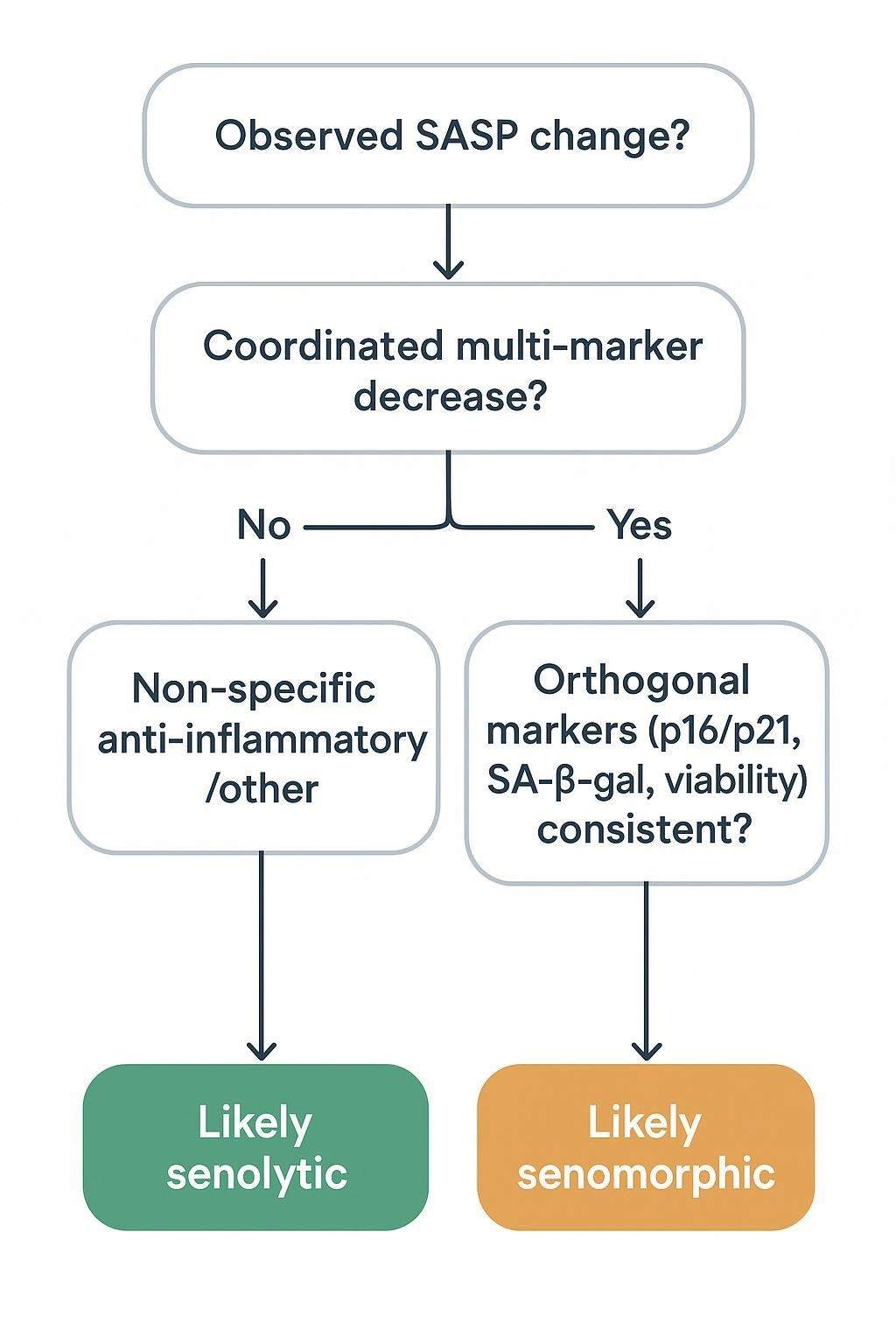
How to Interpret SASP Readouts Correctly
What does a strong SASP result look like? In practice, you're looking for coordinated change across multiple factors within a biologically coherent panel, directional consistency that fits the model and timing, and agreement with orthogonal senescence markers. A reduced SASP can indicate suppressed secretory activity, but on its own it doesn't prove senescent-cell clearance; similar patterns can arise from senomorphic modulation, altered viability, or broader anti-inflammatory effects. Pair panel results with cell-cycle arrest markers, SA-β-gal activity, p16/p21 expression, and viability/cell-number controls—especially in intervention studies.
Analytical and Experimental Design Considerations
Robust SASP data depend on disciplined pre-analytical control, fit-for-purpose assay design, and comparability plans that survive across runs, sites, and time.
Pre-analytical choices that matter include CM collection windows (e.g., early vs mature SASP), serum vs plasma handling differences, freeze–thaw management, and matrix effects. On the assay side, consider the dynamic range across mixed analyte classes, detectability of low- vs high-abundance markers, the balance between panel breadth and reproducibility, and whether you need a focused screening panel or an expanded profiling set. For screens and longitudinal work, cross-run consistency, panel stability, and standardized workflows (including bridging controls) are central to interpretability.
Table — Pre-analytics and validation checklist (map to CLSI EP-series; ICH M10 for LBAs)
| Area | What to document/test | Notes |
|---|---|---|
| Sample handling | Tube type (e.g., EDTA, citrate, CTAD), processing time/temperature, centrifugation, storage, freeze–thaw cycles | Platelet mitigation (CTAD or PPP) for platelet-sensitive analytes; time-to-spin ideally <3–4 h |
| Detection capability | LOD/LOQ per analyte (EP17-A2); confirm ≤20% CV at LLOQ where feasible | Use matrix-matched low-level samples; control α/β risk |
| Precision | Intra-/inter-assay precision (EP05-A3) | Aim for ≤15% CV intra-run and ≤20% inter-run in research contexts when feasible |
| Linearity/parallelism | Dilution linearity and parallelism (EP06-A) | Assess across analytical range; slopes near 0.8–1.25 are common acceptance heuristics |
| Recovery & bias | Spike–recovery and method comparison (EP09-A3/EP15-A3) | Typical targets 80–120% recovery unless justified otherwise |
| Interference & stability | Hemolysis/icterus/lipemia (EP07-A2); bench-top, long-term, and freeze–thaw stability (EP25-A) | Evaluate at relevant decision levels; record acceptance criteria a priori |
Common Pitfalls in SASP Biomarker Studies
A frequent error is treating SASP as a fixed, universal signature. Composition changes across models and tissues, so a single universal panel is rarely optimal. Another pitfall is overconfident blood-based claims: circulating proteins are indirect compared with conditioned-media readouts, and background inflammation or platelet activation can obscure senescence-linked interpretation. It's also easy to confuse anti-inflammatory activity with senolysis; lower cytokines don't necessarily mean fewer senescent cells. Finally, building an overly broad panel without a sharp question often reduces reproducibility; let the experimental decision point drive marker selection.
How to Build a More Useful SASP Resource Framework
Start with the biological question: are you confirming senescence, profiling the secretome, screening senolytics, monitoring senomorphic effects, or looking for circulating correlates? Match panel scope to the sample type—focused secreted-factor sets for conditioned-media work, more conservative backbones for serum or plasma studies, and expanded discovery panels only when justified by the goal. Pair multiplex SASP data with orthogonal evidence: couple secreted proteins to cellular senescence markers, combine functional interpretation with cell-state confirmation, and include viability-aware analysis whenever you interpret intervention effects.
Frequently Asked Questions
What are the most commonly measured SASP biomarkers?
Short answer: a mix of inflammatory cytokines (IL-6, IL-8/CXCL8, IL-1α/β), chemokines (e.g., CCL2/MCP-1), matrix remodelers (MMP-1/3; PAI-1/SERPINE1), and sometimes growth factors (GM-CSF, VEGF). The optimal set depends on your model, matrix, and objective. Panels designed as a coherent whole outperform one-off markers.
Should I measure SASP in conditioned media or in blood (serum/plasma)?
Use conditioned media for mechanistic, cell-autonomous questions and clean comparisons across induced vs control conditions. Choose serum or plasma when studying systemic or translational signals—but apply stricter pre-analytical control (tube, timing, temperature) and interpret conservatively because multiple sources contribute to the readout. Serum and plasma aren't interchangeable; platelet-sensitive targets are better assayed in appropriately prepared plasma.
Can a multiplex SASP panel prove senolytic efficacy?
Not by itself. Treat coordinated SASP decreases as hypothesis-generating. Stronger senolytic claims require orthogonal evidence that senescent-cell burden truly fell—e.g., changes in p16/p21, SA-β-gal, and viability/cell counts consistent with selective clearance, not just suppressed secretion.
How do I know if my multiplex SASP panel result is credible?
Look for a coordinated multi-marker pattern that fits your model and timing, documented pre-analytics, and per-analyte validation (LOD/LOQ, precision, dilution linearity, recovery). Agreement with orthogonal senescence markers is the clincher. If one or two markers drive the signal without panel-level support, be cautious.
Which platform should I start with for a new SASP panel?
If you need moderate-to-high plex with efficient sample use, bead-based multiplexing (e.g., Luminex xMAP) is a common starting point. If a few low-abundance targets are critical, consider Simoa for those specific analytes. For broader discovery, Olink panels can be useful, but confirm key hits on an orthogonal quantitative platform. For planar ECL, MSD often offers strong dynamic range and precision for targeted sets. The best choice depends on your analytes, matrix, and validation needs; it's common to mix platforms across phases.
References:
- Measuring the Senescence-Associated Secretory Phenotype (Biomedicines, 2025): overview of SASP composition and multiparametric protein measurement strategies. Accessible via the open-access article in the Biomedicines collection: https://pmc.ncbi.nlm.nih.gov/articles/PMC12467123/
- Frontiers in Immunology (2020) comparison of bead-based fluorescence (Luminex xMAP) vs planar electrochemiluminescence (MSD) multiplex cytokine assays, discussing sensitivity, dynamic range, and concentration bias in matched plasma/serum: https://www.frontiersin.org/articles/10.3389/fimmu.2020.572634/full
- JCI Insight (2020) work on circulating SASP and medical risk, illustrating matrix remodelers and inflammation in translational cohorts: https://insight.jci.org/articles/view/133668
- PLOS Biology review (2023) on senescence, SASP dynamics, and context dependence, including temporal evolution considerations: https://journals.plos.org/plosbiology/article?id=10.1371%2Fjournal.pbio.3002326
- Frontiers in Immunology (2024) discussion of pre-analytical pitfalls for cytokine measurement, including handling delays and temperature control: https://www.frontiersin.org/articles/10.3389/fimmu.2024.1382192/full
- PLoS One (2012) evidence that CTAD tubes reduce platelet-derived protein artifacts in plasma, with quantitative examples for PDGF: https://journals.plos.org/plosone/article?id=10.1371%2Fjournal.pone.0038290
- PMC (2023) platelet-derived soluble CD40L review highlighting platelet cargo contributions to measured biomarkers: https://pmc.ncbi.nlm.nih.gov/articles/PMC10002540/
- Aging Cell (2024) evidence for ECM remodeling and MMP activity in senescence-associated tissue changes: https://pmc.ncbi.nlm.nih.gov/articles/PMC11561669/
- CLSI Standards (EP05-A3, EP06-A, EP07-A2, EP09-A3, EP15-A3, EP17-A2, EP25-A) index for precision, linearity, interference, method comparison, detection capability, and stability studies: https://clsi.org/shop/standards/
- ICH M10 guideline on bioanalytical method validation for ligand-binding assays, covering LLOQ definitions, accuracy/precision targets, and stability: https://ich.org

