Anti-aging interventions are often easier to describe than to measure. In healthy or only mildly inflamed cohorts, biological changes after NMN, exercise, or nutritional supplements are usually modest, variable, and slower-moving than changes in symptoms or performance. That is why a useful biomarker strategy should focus less on proving a broad "anti-aging effect" and more on detecting whether an intervention shifts low-grade inflammatory biology in a repeatable, interpretable way. An inflammaging panel provides that structure by combining cytokines, chemokines, and systemic anchors into a shared backbone that can be followed across interventions and timepoints, with limited mechanism-specific additions when justified.
This guide is designed to help you do that more rigorously: choose a practical core marker set, decide when to expand it, plan sampling around the biology of each intervention, and compare NMN, exercise, and supplement arms without forcing them into the same interpretive model. The goal is not to turn a biomarker panel into proof of slowed aging, but to generate a more reliable picture of whether an intervention is changing inflammaging-related biology over time.
How to Measure Inflammaging in Intervention Studies
In anti-aging intervention studies, the main challenge is often not whether an intervention works, but whether the biological effect is large enough to detect. Many cohorts are healthy or only mildly inflamed, so changes after NMN, exercise, or supplements are often subtle. A single marker can miss that signal—or make a small fluctuation look more important than it is.
That is why inflammaging is a useful measurement framework. Instead of relying on one analyte, it looks at low-grade inflammatory biology as a pattern. A practical panel may include cytokines such as IL-6, TNF-α, and IL-1β, chemokines such as IL-8/CXCL8, MCP-1/CCL2, and CXCL10, plus adjacent markers such as hsCRP. Read over time, these markers can show whether an intervention is shifting inflammatory tone in a coherent way.
This page is designed to help you decide which biomarkers to include, how broad the panel should be, when to collect samples, and how to interpret change without overstating anti-aging effects.
What to Measure in an Inflammaging Biomarker Panel
Core pro-inflammatory cytokines
A practical starting point includes IL-6, TNF-α, IL-1β, and—where relevant—IL-18. These markers represent central pro-inflammatory axes that can register small shifts in low-grade inflammation. IL-6, in particular, is frequently detectable and informative in older adults, but it should not be used in isolation. TNF-α and IL-1β provide complementary context about innate inflammatory tone, while IL-18 can add value in metabolic-inflammation or inflammasome-linked designs. In healthy cohorts, some of these analytes may approach the lower limit of quantitation (LLOQ) on standard multiplex platforms, so platform selection and pre-study detectability checks are essential.
Chemokines associated with immune-cell recruitment
MCP-1/CCL2 and IL-8/CXCL8 are often more readily detectable than ultra-low cytokines and can reflect monocyte trafficking and neutrophil/epithelial signaling, respectively. CXCL10 (IFN-γ–inducible protein-10) is useful in broader immune-aging panels and NAD- or viral-pathway contexts because it reflects interferon-axis activity. Chemokines add value beyond classic cytokines by capturing recruitment signals and interferon-related dynamics that may change with training status, metabolic health, or supplement mechanisms.
Regulatory and counter-inflammatory markers
IL-10 and IL-1RA are canonical counter-regulatory signals. TGF-β, in expanded study designs, may inform tissue remodeling and regulatory balance. In many anti-aging contexts, pattern and balance matter more than any single "anti-inflammatory" marker: for example, a small IL-6 decrease accompanied by a coherent shift in MCP-1 and a stable or context-appropriate IL-10 behavior is more credible than an isolated IL-6 dip.
Broader inflammaging-adjacent proteins
hsCRP serves as a systemic anchor with strong epidemiological context; it is less specific than cytokines but improves interpretability. Soluble receptors (e.g., TNFR1) can increase robustness and are common in senescence-adjacent designs. SASP-linked proteins (e.g., GDF15, HMGB1) may be appropriate when your study probes senescence biology. If your intervention targets energy metabolism or muscle adaptation, consider metabolic–inflammatory markers that clarify context (e.g., adiponectin/leptin in metabolic arms), while keeping the core inflammatory readouts stable for comparability.
To keep the scope actionable, many teams begin with a shared 6–8-analyte backbone and add 2–4 mechanism-specific markers as needed.
| Marker group | Example analytes | Why they matter in low-grade cohorts |
|---|---|---|
| Pro-inflammatory cytokines | IL-6, TNF-α, IL-1β, IL-18 (context) | Core innate inflammatory tone; small but interpretable shifts in longitudinal designs |
| Chemokines | MCP-1/CCL2, IL-8/CXCL8, CXCL10 | Recruitment and interferon-axis signals; often better detectability |
| Counter-regulatory | IL-10, IL-1RA; TGF-β (expanded) | Balance and pattern interpretation; avoid over-reading single "anti-inflammatory" markers |
| Systemic anchors | hsCRP; soluble receptors (e.g., TNFR1) | Contextualize cytokine movement; improve panel-level robustness |
| SASP/mechanistic add-ons | GDF15, HMGB1 (context) | Link senescence/inflammaging when hypothesis-driven |
Which Biomarkers Are Most Relevant by Intervention Type
NMN and NAD-related intervention studies
For NMN or NAD-adjacent interventions, pair the shared backbone with markers interpretable alongside NAD or metabolic readouts. Keep the core stable if the study includes NMN-only and combination arms so you can compare effects on a common scale. Because circulating cytokine changes may be modest, favor analytes with good detectability (e.g., IL-6, MCP-1, IL-8) and consider CXCL10 if your hypothesis touches interferon signaling. Treat any observed systemic cytokine movement as exploratory unless corroborated by metabolic or NAD-pathway shifts.
Exercise intervention studies
Exercise produces time-sensitive immune responses. Distinguish acute, bout-related cytokine transients from chronic training adaptations. The shared backbone remains valid, but sampling and interpretation rules differ: immediate post-exercise increases in IL-6 or IL-10 do not necessarily indicate worsened inflammaging. Maintain resting-state longitudinal draws for adaptation, and reserve separate acute windows if you want to analyze bout kinetics. Choose markers that remain interpretable across aerobic, resistance, and mixed protocols.
Supplement and nutraceutical studies
Supplements span heterogeneous mechanisms—from omega-3 fatty acids to polyphenols, probiotics, and botanicals—so the backbone is critical. Favor markers with high detectability and reproducibility in small- to mid-sized cohorts. Add mechanisms selectively (e.g., include CXCL10 if an interferon-modulating botanical is under test). Use the same core across arms to compare effect sizes without overfitting to one product's putative pathway.
Combination intervention studies
For multi-arm designs that mix NMN, exercise, and supplements, use a shared backbone across all arms and add mechanistic markers sparingly. Define upfront whether the study seeks additive effects, synergy, or a comparative ranking, because that decision determines the needed statistical power and sampling cadence.
How to Choose Between a Focused Panel and an Expanded Panel
When a focused panel is the better choice
Focused panels shine when you need repeated measures across many timepoints, have small or mid-sized cohorts, or prioritize effect tracking over pathway discovery. A compact set reduces non-detects, simplifies QC, and preserves power for longitudinal statistics. It also stabilizes cross-arm comparability, which is essential if you intend to compare NMN, exercise, and supplement effects.
When an expanded panel makes sense
Use expanded panels in exploratory pilots, screens with multiple candidate interventions, or mechanistic programs that explicitly link inflammatory outcomes to broader aging biology (frailty, resilience, or senescence-associated pathways). Expanded designs benefit from pre-screening detectability and a clear hypothesis to avoid diluting interpretability.
How to avoid panel bloat
Cull analytes with poor detectability, remove markers that are hard to interpret across intervention classes, and preserve a high-value core across all waves. Add mechanistic targets selectively rather than inflating the entire panel by default. Think of add-ons as modular tiles plugged into a fixed backbone, not a reason to rebuild your core each time.
| Scenario | Panel scope | Rationale |
|---|---|---|
| Longitudinal response tracking in small cohorts | Focused (6–12 analytes) | Maximizes detectability and precision; reduces QC burden |
| Multi-arm exploratory screen | Expanded (20–60 analytes) | Broader hypothesis space; requires pre-screening and careful statistics |
| Cross-arm comparability study | Shared core + 2–4 add-ons | Keeps results on a common scale; add-ons only where mechanism demands |
When to Collect Samples in Anti-Aging Intervention Studies
Baseline sampling
Collect pre-intervention samples under standardized conditions. Match fasting status, recent physical activity, and timing relative to supplement intake. If feasible, perform morning draws to control for diurnal variation. Document baseline conditions clearly to enable clean longitudinal comparisons.
Early response windows
If your question is, "Do we see an early biological signal?" add short-term timepoints. These are particularly relevant for NMN/NAD-related signaling or exercise-related immune kinetics. Keep these windows analytically separate from adaptation-focused endpoints to avoid interpretive cross-talk.
Midpoint and endpoint sampling
For gradual effects, rely on multiple longitudinal timepoints (e.g., baseline, mid, and end) rather than a single post-intervention snapshot. Repeated measures increase credibility and help differentiate noise from signal in healthy or lightly inflamed populations. Keep timing consistent across all arms.
Special timing rules for exercise studies
Create a branch for acute sampling around exercise bouts (immediate post, +5 min, +2–3 h, +4–6 h, +24 h, +72 h) separate from resting-state longitudinal draws. Do not mix these in the same interpretive layer. Align blood collection with exercise scheduling and recovery windows so that bout kinetics do not contaminate adaptation trends.
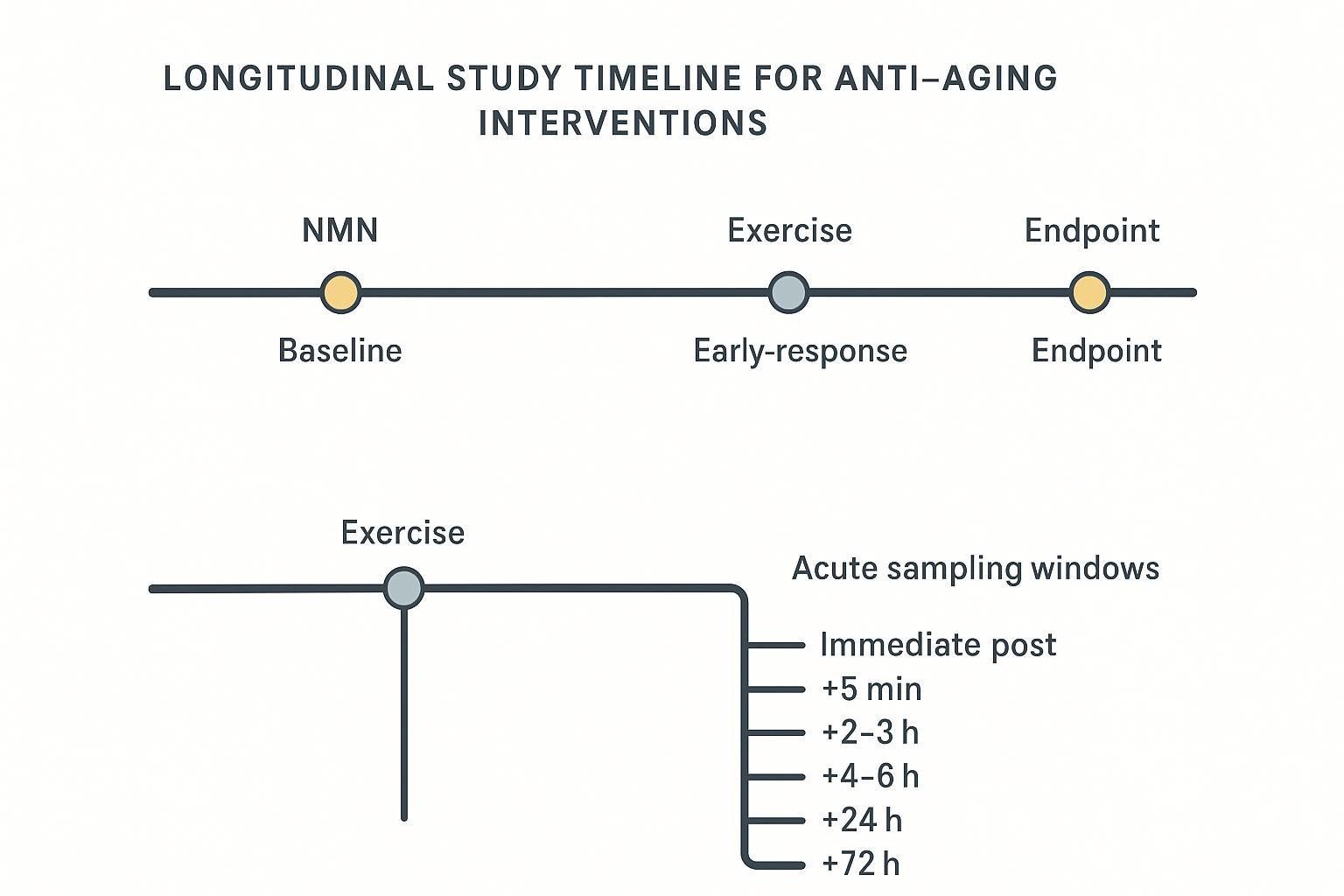
| Intervention type | Resting-state longitudinal timing | Acute/early-response timing (if applicable) |
|---|---|---|
| NMN/NAD-related | Baseline; early (1–2 weeks); mid (4–8 weeks); end (8–12+ weeks) | Optional early windows for signaling checks; avoid conflating with adaptation |
| Exercise training | Baseline; mid (4–8 weeks); end (8–12+ weeks), time-of-day matched | Pre; immediate post; +5 min; +2–3 h; +4–6 h; +24 h; +72 h |
| Supplements | Baseline; early (1–2 weeks); mid (4–8 weeks); end (8–12+ weeks) | Usually resting-state only; acute windows rare unless mechanism suggests |
How to Read Inflammaging Biomarker Changes Without Overclaiming
Read changes as coordinated patterns
A single reduced cytokine is rarely enough to warrant strong anti-aging claims. Pattern-level movement across a defined set—e.g., coherent shifts in IL-6, MCP-1, and hsCRP—speaks more convincingly than isolated wins. Concordant trajectories across two or more markers strengthen the argument for intervention-related biology.
Distinguish measurable change from meaningful change
Statistical detectability in a small cohort does not guarantee biological significance. Interpretation improves when inflammatory shifts align with functional performance, metabolic endpoints, or training-load context. In healthy cohorts, small within-range changes should be framed cautiously and never treated as proof of slowed biological aging.
Account for intervention-specific biology
Transient post-exercise cytokine rises are part of normal adaptation and do not indicate worsening inflammaging. NMN-related effects may be indirect or modest; read them alongside NAD or metabolic measures. Supplement responses vary by mechanism and dose; avoid oversimplifying heterogeneous formulations into one category.
Favor longitudinal interpretation
Repeated trajectories are more credible than single pre/post contrasts. Stable directional change across timepoints is especially important when effect sizes are small. Longitudinal consistency also improves comparability across NMN, exercise, and supplement arms.
How to Compare NMN, Exercise, and Supplement Effects Fairly
Use a shared core biomarker backbone
Keep the most informative inflammatory markers consistent across all arms and timepoints. Add intervention-specific markers only when the mechanism requires them. This preserves your ability to compare magnitudes and directions on a common biological scale.
Normalize expectations by intervention class
Plan for time-sensitive behavior in exercise studies; consider adjacent NAD/metabolic context in NMN studies; and subgroup supplements by mechanism rather than treating them as a monolith. Calibrating expectations up front prevents over-interpretation of small or short-lived shifts.
Avoid false equivalence across studies
Ages, baseline inflammatory burden, body composition, training status, and assay design all affect observed patterns. Cross-study comparisons should rely on shared markers, comparable sampling logic, and matched interpretation rules, not superficial ranking.
How to Build a Stronger Inflammaging Study Design
Start with the intervention question
Decide whether the primary goal is to detect a response, compare arms, rank candidates, or support a mechanism claim. Clarify whether your study is exploratory, confirmatory, or focused on longitudinal monitoring. Let these choices drive panel scope, timing, and statistics.
Match the panel to the study population
Healthy adults, older adults with low-grade inflammatory burden, trained versus sedentary cohorts, and supplement users with heterogeneous baseline status each pose different detectability and interpretation realities. Cohort biology determines how many non-detects you will see and how stable your trends will be; plan accordingly.
Build around repeatability
Choose analytes that are measurable across your cohort and remain interpretable at multiple timepoints. Keep the core backbone stable across phases, and avoid unnecessary analyte turnover once the study begins. This is where multiplex platforms can help by conserving volume while maintaining consistent readouts over time. When ultra-low abundance is a concern, consider an ultrasensitive single-/low-plex for a small subset of targets while retaining the multiplex core.
Pair inflammatory markers with adjacent outcome layers
Improve interpretability by aligning inflammatory signals with functional performance, body composition and training-load context, metabolic outcomes, and—where relevant—NAD readouts in NMN studies. Broader aging measures can be added where they materially clarify conclusions.
Practical note: For longitudinal shared backbones with repeated measures, many groups opt for a Luminex-first workflow to maximize plex size and conserve volume, then layer a targeted ultrasensitive assay for the 1–2 lowest-abundance cytokines.
In practical terms, service providers can facilitate this hybrid approach. For example, a Luminex multiplex assay can support high-plex, low-volume longitudinal profiling, while a Simoa cytokine assay can target the most challenging low-abundance analytes.
Analytical and Workflow Considerations
Sample type and collection consistency
Pick serum or plasma deliberately; report anticoagulants if using plasma. Standardize time-of-day, fasting status, and recent activity. Minimize pre-analytical variability: process within 30–60 minutes when possible, document storage conditions, and control freeze–thaw cycles consistently across timepoints.
Platform and assay design considerations
For longitudinal, low-volume studies, multiplex suitability matters as much as raw sensitivity. Luminex supports high plex and efficient volume use, but very low-abundance cytokines (e.g., IL-1β) may need ultrasensitive approaches. MSD V-Plex offers a balanced option; MSD S-Plex and Simoa push sensitivity further with lower plex. Olink enables very high plex with minimal volume in normalized units and strong specificity, which can be attractive for broader discovery while maintaining a compact focused subset for tracking.
Cohort and background-noise considerations
Healthy aging cohorts often show weaker separation than disease cohorts, and baseline variability can obscure modest effects. Anticipate small changes and plan repeats. Favor mixed-effects models for longitudinal data and avoid imputing non-detects without justification.
| Platform | Sensitivity for very low cytokines | Plex size | Sample volume | Practical note |
|---|---|---|---|---|
| Simoa | Very high (fg/mL-level) | Low–moderate | Low–moderate | Best for ultra-low targets; pair with multiplex core for efficiency |
| MSD S-Plex | Very high | Low–moderate | Moderate | Strong low-end detection; use for a few critical analytes |
| MSD V-Plex | Moderate | Moderate | Moderate | Balanced option when ultra-low sensitivity is not essential |
| Luminex xMAP | Moderate (pg/mL) | High (50–100) | Low (≈25–50 µL) | Efficient for shared backbones; verify detectability per analyte |
| Olink PEA | Moderate–high; high specificity | High (48–96) | Very low (≈1–2 µL) | Normalized units; excellent for discovery with focused tracking set |
Detectability and QC checklist (fit-for-purpose)
- Pre-screen detectability for low-abundance cytokines in your matrix/platform.
- Validate LLOQ, precision (≤15–20% CV targets), dilution linearity/parallelism, recovery/matrix effects, and stability per analyte.
- Control cross-reactivity in high-plex assays; confirm critical markers via orthogonal methods if needed.
- Lock the core backbone before longitudinal enrollment; limit mid-study changes.
Common Pitfalls in Anti-Aging Biomarker Studies
Using biomarkers that are too broad or too nonspecific
Relying solely on broad systemic markers can miss intervention-relevant biology. Tie selection to study logic, and combine systemic anchors with mechanistically informative cytokines and chemokines.
Over-claiming anti-aging effects from short-term biomarker shifts
Early movement, especially within reference ranges, rarely proves slowed biological aging. Seek longitudinal consistency and alignment with adjacent outcomes before making strong claims.
Mixing acute and chronic biology
This is especially problematic in exercise or combination arms. Separate immediate post-bout sampling from resting-state adaptation windows in design and analysis.
Designing around trend language instead of measurement logic
"Longevity biomarkers" is too vague to guide assays. Start from the intervention class, study population, panel scope, and timing strategy needed to answer an explicit question.
FAQ
Which biomarkers are most useful for measuring inflammaging in intervention studies?
A focused set of repeatable cytokines (IL-6, TNF-α, IL-1β), chemokines (MCP-1/CCL2, IL-8/CXCL8, CXCL10), and systemic anchors (hsCRP) forms a practical core. Add 2–4 mechanistic markers (e.g., TNFR1, GDF15) only when your hypothesis demands them.
Can one biomarker panel work across NMN, exercise, and supplement studies?
Yes—when you use a shared core for comparability and add a few targeted markers by mechanism. It does not work when every arm simultaneously requires deep profiling of distinct pathways.
What makes an anti-aging biomarker result more credible?
Clear baseline definition, appropriate sampling windows, longitudinal trend analysis, pattern-based interpretation, and alignment with functional or metabolic outcomes. Use mixed-effects models and pre-specified interpretation rules.
How should exercise biomarker timing differ from supplement biomarker timing?
Exercise often needs both acute (pre, immediate post, +5 min, +2–6 h, +24–72 h) and chronic resting-state draws (weeks to months). Supplements typically rely on resting-state longitudinal comparisons at baseline, early, mid, and end.
References:
- Sayed N, et al. An inflammatory aging clock (iAge) based on deep learning tracks multimorbidity, immunosenescence, frailty and cardiovascular aging. Nat Aging. 2021. https://pubmed.ncbi.nlm.nih.gov/34888528/
- Verberk IMW, et al. Pre-analytical sample handling effects on blood cytokine levels. 2021. https://pubmed.ncbi.nlm.nih.gov/34289718/
- Goj T, et al. Acute Cytokine Response to 30-Minute Exercise Bouts Before and After Training. J Clin Endocrinol Metab. 2023. https://academic.oup.com/jcem/article/108/4/865/6774023
- de Sousa CAZ, et al. Time Course and Role of Exercise-Induced Cytokines in Muscle Damage and Repair after a Marathon Race. Front Physiol. 2021. https://www.frontiersin.org/articles/10.3389/fphys.2021.752144/full
- Pillon NJ, et al. Distinctive exercise-induced inflammatory response and exerkines. Sci Adv. 2022. https://www.science.org/doi/10.1126/sciadv.abo3192
- McKinski K, et al. Comparison of highly sensitive, multiplex immunoassay platforms. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11749433/
- Günther A, et al. Comparison of bead-based fluorescence vs planar array multiplex immunoassays. Front Immunol. 2020. https://www.frontiersin.org/articles/10.3389/fimmu.2020.572634/full
- Basisty N, et al. A proteomic atlas of senescence-associated secretomes. PLoS Biol. 2020. https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.3000599
- Teissier T, et al. Interconnections between inflammageing and cellular senescence. 2022. https://pmc.ncbi.nlm.nih.gov/articles/PMC8834134/
- Frontiers in Immunology Review (2024): IL-6 utility in older adults and inflammaging overview. https://pmc.ncbi.nlm.nih.gov/articles/PMC10943692/

