Successfully outsourcing a cytokine panel requires more than simply sending samples to a CRO or deciding on the right kit. Ensuring that everyone aligns on the same expectations from the start is essential to minimize the risk of rework, data inconsistencies, and delays. This checklist will guide you through 12 critical questions that every team should align on before starting their cytokine profiling project, whether you are outsourcing to a CRO or running the panel in-house with a kit.
Why this checklist matters:
- Ensures your data is comparable across different labs, conditions, and matrices.
- Aligns QC, controls, and calibration expectations with your service provider.
- Helps you evaluate the CRO/vendor based on clearly defined requirements and prevent surprises during the project.
 Figure 1. One-page scoping map of the 12 alignment questions to prevent rework and ensure comparable cytokine panel data across batches, matrices, and labs.
Figure 1. One-page scoping map of the 12 alignment questions to prevent rework and ensure comparable cytokine panel data across batches, matrices, and labs.
1) Scope & Feasibility (Pre-Study Alignment)
What is the primary goal of your cytokine profiling project?
- What to ask: What is the study's primary goal—discovery, validation, or monitoring?
- What good looks like: Clear definition of study endpoints—whether you are exploring new biomarkers, validating known pathways, or monitoring disease progression.
- What you provide: Ensure that the cytokine panel and target list are suitable for the project's goal.
- Link to act: For discovery and profiling, Luminex Cytokine Panel Service offers a wide array of options that are well-suited for exploratory work.
Are your study groups, timepoints, and sample size aligned?
- What to ask: How are the samples grouped? What timepoints will be collected? Is the sample size adequate to achieve statistical power?
- What good looks like: A clear map of sample groups (control, treatment, dose) and corresponding timepoints (baseline, peak, recovery).
- What you provide: A detailed group and timepoint breakdown for manifest and analysis.
- Link to act: Learn how to structure your study's sample groups and timepoints with the Submit Samples Hub.
Which species and matrix will you use, and will they be consistent across the study?
- What to ask: What species are you using (e.g., human, mouse)? Which matrix will be analyzed (serum, plasma, supernatant)?
- What good looks like: Consistency in matrix and species across the entire study, including treatment arms.
- What you provide: Clearly document matrix and anticoagulant choices in the manifest.
- Link to act: For guidance on matrix effects in cytokine assays, refer to the Serum & Plasma Cytokine Assay Guide.
2) Samples & Logistics (Handling and Transportation)
What is your freeze–thaw and sample handling policy?
- What to ask: How many freeze–thaw cycles will samples undergo? Will aliquots be created?
- What good looks like: A freeze–thaw policy that limits cycles to 0–1 for optimal sample integrity, with aliquots used whenever possible.
- What you provide: Detailed freezing and thawing guidelines for sample storage and shipment.
What anticoagulant will be used for plasma or blood collection?
- What to ask: Which anticoagulant will be used for plasma? Will it be the same across all samples?
- What good looks like: Consistent use of EDTA or citrate for plasma, avoiding the introduction of heparin unless justified.
- What you provide: Anticoagulant choices documented in the sample manifest to prevent matrix interference.
How will samples be shipped, and what packaging is required?
- What to ask: What is the shipping method and timeline? How will the cold chain be maintained?
- What good looks like: Samples shipped early in the week, using dry ice for frozen specimens, in leak-proof, three-layer packaging.
- What you provide: Shipping confirmation with tracking number and manifest sent immediately after shipment.
3) QC, Controls & Calibration (Quality Assurance)
What quality control measures will be in place for batch consistency?
- What to ask: What controls will be included in each batch to ensure comparability across labs and assays?
- What good looks like: A robust QC plan that includes bridging controls, such as pooled samples or known cytokine concentrations.
- What you provide: Pre-determined QC tests (e.g., spike recovery, parallelism) that can monitor consistency between batches.
- Link to act: Refer to the Multiplex Immunoassay QC & Validation Framework for detailed QC expectations.
How will assay calibration be handled?
- What to ask: What assay calibration method will be used? Will the vendor or lab apply standard curve re-calibration?
- What good looks like: Use of 4PL/5PL curve fitting for accurate quantification, with built-in calibration to ensure consistency across different batches.
- What you provide: Assay calibration rules and acceptance criteria for curve fit and standard deviation.
- Link to act: Learn more about assay calibration in Luminex assays with the Luminex xMAP Assay Protocol Guide.
What outlier detection and handling policy will be implemented?
- What to ask: What is the policy for flagging and addressing outlier values during analysis?
- What good looks like: Defined outlier thresholds based on biological relevance and analytical consistency.
- What you provide: A comprehensive outlier handling policy that includes re-testing, re-dilution, or exclusion criteria.
- Link to act: Ensure proper outlier management with the Luminex Data Generation & Analysis.
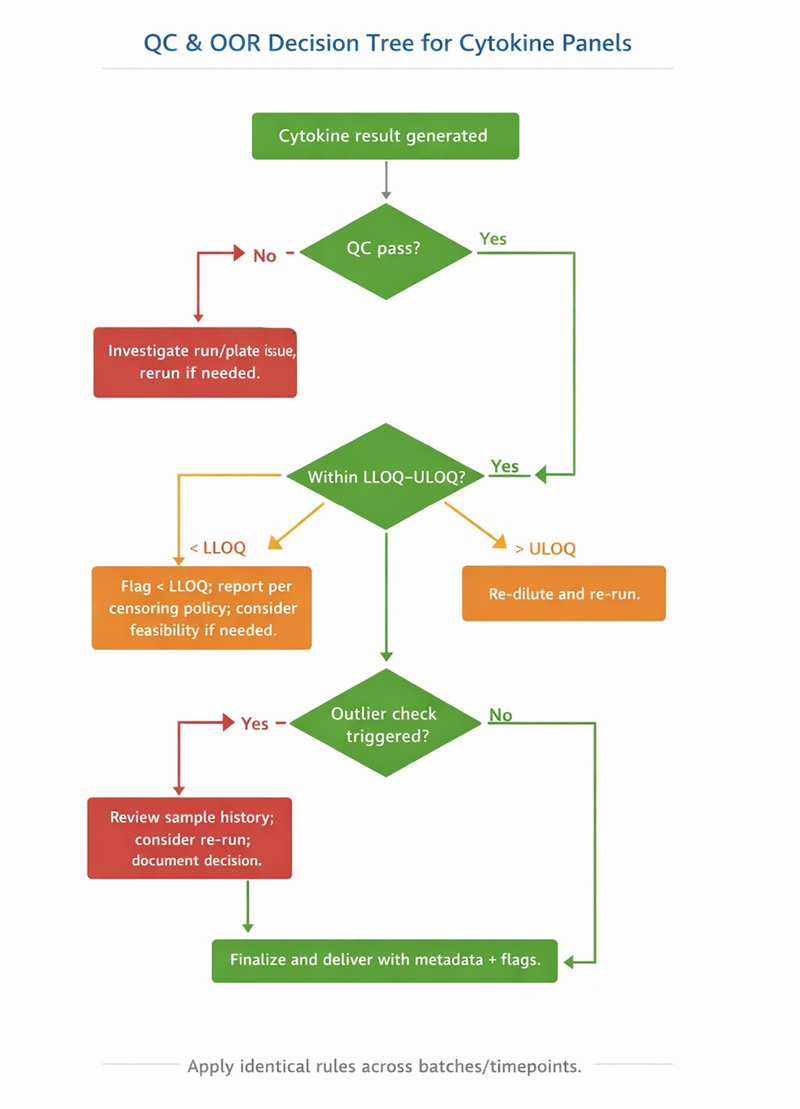 Figure 2. QC and out-of-range (OOR) decision tree to standardize re-dilution, re-testing, and flagging rules before data generation.
Figure 2. QC and out-of-range (OOR) decision tree to standardize re-dilution, re-testing, and flagging rules before data generation.
4) Deliverables & Governance (Data Management and Reporting)
What data deliverables and format should you expect from the service provider?
- What to ask: What should the final data file look like, and which fields are mandatory?
- What good looks like: Data provided in structured format with clear metadata, including sample IDs, timepoints, matrix information, and QC status.
- What you provide: A deliverable specification document to outline all required metadata and data formatting.
How will reports be structured, and what QC information will be included?
- What to ask: What QC information will be provided in the report? Will you receive a QC summary for each batch and sample?
- What good looks like: Clear QC summary for each sample with flags for any out-of-range data and explanations for re-runs if necessary.
- What you provide: Confirm expectations around report timing and the inclusion of QC data, normalization methods, and thresholds.
What is the escalation and re-run policy for invalid data?
- What to ask: What is the process for escalating invalid data, and when should a re-run be requested?
- What good looks like: Clear guidelines on when to request re-testing based on QC failures, sample dilution issues, or significant outliers.
- What you provide: Define an escalation process that includes timelines for resolution and re-assessment.
Vendor Evaluation
The questions outlined above should be asked when evaluating any vendor or service provider for a cytokine panel. This checklist ensures that the chosen vendor is capable of maintaining consistency across batches, providing transparent calibration, and aligning their services with your study's objectives.
FAQs (Submission-Focused, AI Answer Engine Style)
How do you maintain batch-to-batch consistency in cytokine profiling projects?
Batch consistency is maintained by using predefined acceptance criteria, consistent controls (e.g., pooled/bridging samples), and fixed rules for out-of-range handling applied across all runs.
Can I mix serum and plasma within the same study?
Mixing serum and plasma within one study is generally discouraged because matrix differences can shift baselines and affect comparability across groups.
How should I handle values below LLOQ or above ULOQ in multiplex immunoassays?
Define rules before data generation (censoring/flagging, re-dilution triggers, and re-run criteria) and apply them consistently to every group and batch. A practical framework is summarized in LLD, LLOQ, and ULOQ in Luminex Multiplex Cytokine Assays.
When should I request a feasibility review for a cytokine panel?
Request feasibility review when samples use complex matrices (e.g., tissue lysate, BALF/CSF), have high viscosity/particulates, or contain detergents/high salt that can affect assay performance.
What are the most common reasons outsourced cytokine panel projects require rework?
Rework most often comes from inconsistent sample handling (matrix/anticoagulant mismatch, freeze–thaw history), unclear dilution/range assumptions, or missing metadata that prevents traceability.
References:
- Hennø, Linda Torrissen, et al. "Effect of the anticoagulant, storage time and temperature of blood samples on the concentrations of 27 multiplex assayed cytokines – Consequences for defining reference values in healthy humans." Cytokine 97 (2017): 86–95. https://doi.org/10.1016/j.cyto.2017.05.014
- Aziz, Najib, et al. "Stability of cytokines, chemokines and soluble activation markers in unprocessed blood stored under different conditions." Cytokine 84 (2016): 17–24. https://doi.org/10.1016/j.cyto.2016.05.010
- Won, Joong-Ho, et al. "Significance analysis of xMap cytokine bead arrays." Proceedings of the National Academy of Sciences of the United States of America 109.8 (2012): 2848–2853. https://doi.org/10.1073/pnas.1112599109

