Why This Question Matters in Real Cytokine Studies
The issue is not washing alone, but execution consistency
In bead-based multiplex cytokine assays, washing directly influences bead retention, background control, and ultimately signal stability. The problem is that small differences in timing, aspiration technique, and resuspension discipline can look minor in a single plate—then turn into a real data-quality issue as you scale across more plates, operators, or batches. That's why the shift from manual to automated magnetic washing is best understood as a change in execution control, not just a convenience upgrade.
Why this matters especially in Luminex workflows
Luminex assays depend on consistent magnetic bead handling across repeated wash cycles. When washing varies, you're more likely to see bead loss, variable acquisition quality, and avoidable reruns. In larger studies—or studies trying to resolve smaller biological differences—the washing method can meaningfully influence whether results stay reproducible and operationally scalable.
This guide explains what changes (and what doesn't) when washing becomes automated, which study risks are most affected by washing method, when automated magnetic washing typically adds real value, and when manual washing can still be fit for purpose.
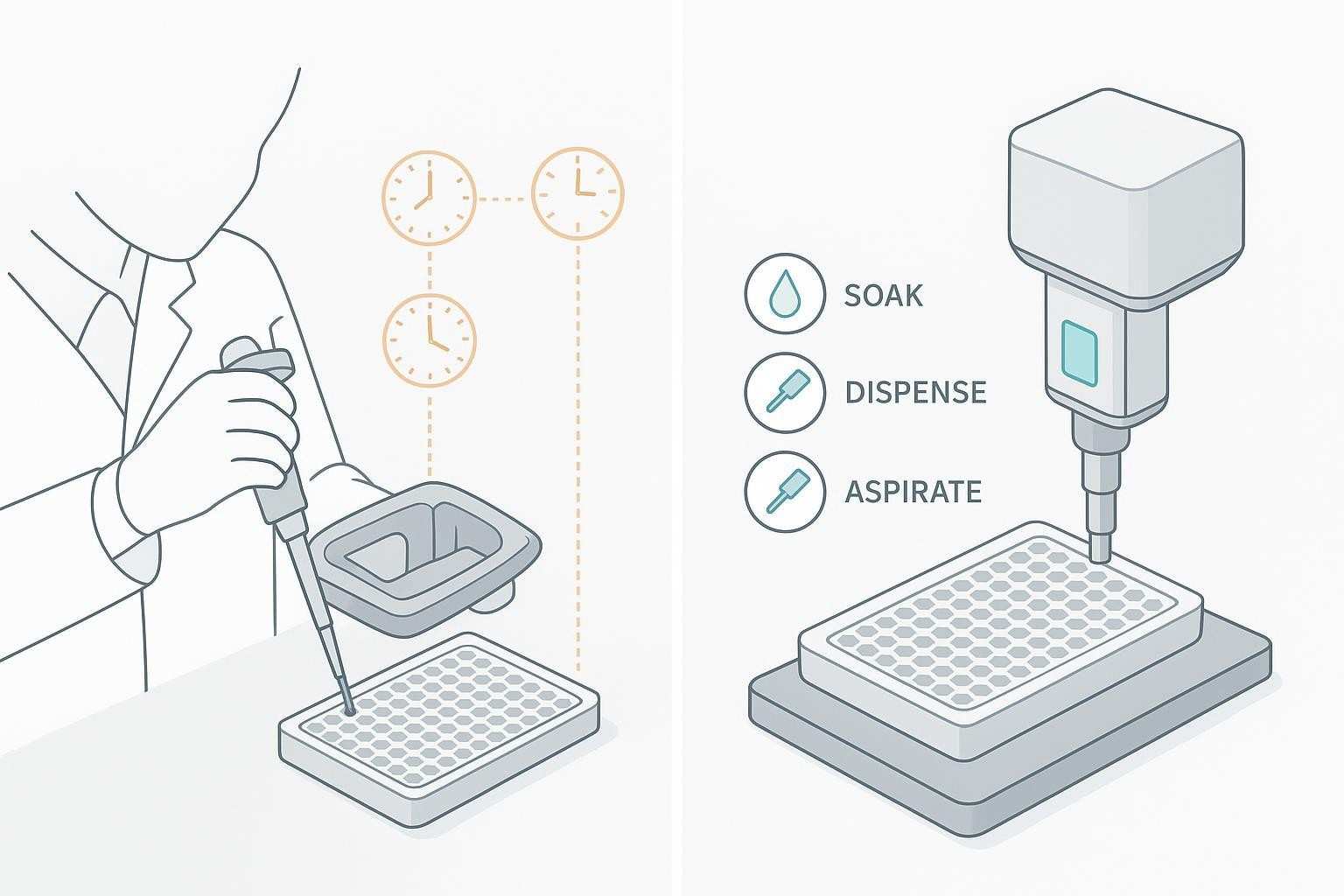
What Actually Changes When Washing Becomes Automated
The shift from operator-dependent handling to standardized wash cycles
With manual washing, results depend heavily on the technician: timing between steps, aspiration consistency, and how repeatably each wash is executed across an entire plate. Automated magnetic washing, by contrast, standardizes bead capture, aspiration, dispensing, and cycle repetition. The main practical change is reduced process variability in a workflow step that is both repetitive and sensitivity-relevant.
What does not change
Automation doesn't change what the panel measures, and it won't rescue an assay that's poorly matched to the study. Matrix effects, analyte abundance, pre-analytical sample quality, and study design still drive a large share of performance outcomes. In other words, automation improves execution control; it doesn't fix fundamental assay fit problems.
Why this distinction matters
It's more accurate to treat automated washing as one component of robust assay execution. It tightens control over a specific step, rather than acting as a universal solution to every source of multiplex assay variability.
Evidence context: Multi‑site programs in Luminex testing have shown that paramagnetic bead use and standardized protocols improve robustness; automation helps enforce repeatability in wash steps rather than acting as a cure‑all. See the multi‑laboratory analysis discussed in the EQAPOL proficiency program, which details sources of variability and the value of standardized methods in Luminex cytokine assays (Rountree et al., 2024).
Using the primary lens: manual vs automated washing in multiplex cytokine assays
- This comparison focuses on how programmed magnetic cycles reduce operator variance relative to hand-executed steps—key to magnetic bead washing reproducibility in scaled studies.
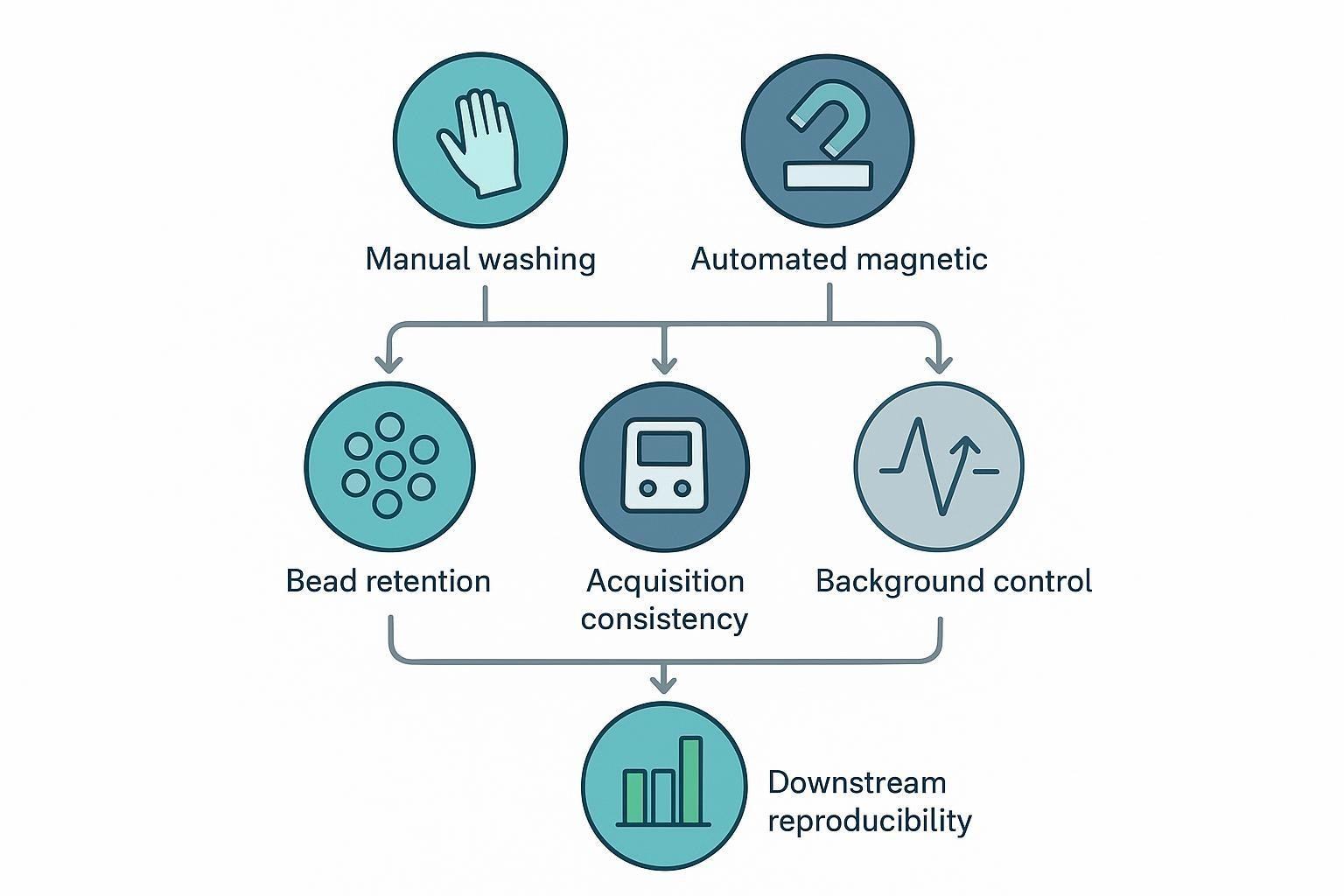
Why Washing Method Can Influence Reproducibility
Bead retention and well-to-well consistency
Reliable bead recovery is a prerequisite for stable signal acquisition across wells and plates. When wash handling varies, bead loss and residual background can become uneven, which widens well-to-well spread and makes plate-level QC harder to interpret. More standardized magnetic washing helps reduce avoidable variability because capture and aspiration are performed the same way, every time.
Operator dependence in repetitive workflows
Manual washing can work well, but performance often varies with technician experience, fatigue, and how many plates must be processed back-to-back. Repetition creates more chances for small execution differences to accumulate into visible CV drift. Automation reduces reliance on person-to-person consistency for one of the most repetitive parts of the assay.
Why reproducibility matters differently at different study scales
A small exploratory run may tolerate modest execution variation. A multi-plate biomarker study is more exposed to handling drift across time, lots, or operators. Reproducibility becomes a higher-stakes requirement when smaller biological differences must remain interpretable instead of being masked by technical spread.
How Washing Method Affects Failure Risk and Rerun Burden
Where wash-related problems tend to appear
Wash-linked issues usually show up as low bead counts, uneven well handling, higher background caused by incomplete or inconsistent washing, or plate-level inconsistency that makes results harder to interpret with confidence.
Why these problems matter more than they first appear
Wash-related failures don't just create a few "bad wells." They increase rerun burden—consuming time, reagents, and often valuable sample volume. In precious-sample or longitudinal studies, avoidable reruns can directly reduce data completeness and push timelines.
How automation changes the risk profile
By making wash cycles more consistent, automation can reduce handling-related variability and therefore reduce some avoidable reruns. The benefit is typically most visible in repetitive, multi-plate, or higher-throughput workflows where small execution differences have more chances to accumulate.
Practical note: Manufacturer application notes for magnetic programs on automated washers describe standardized soak/aspirate cycles that help retain beads and minimize residuals—mechanisms that plausibly reduce rerun triggers when runs scale.
Throughput Is Only Part of the Story
Faster processing is not the only reason automation matters
Throughput gains are real, but speed alone isn't the deciding factor in many biomarker studies. The larger value of automation is repeatable execution under production-like conditions. A workflow that's fast but inconsistent doesn't scale well—and it often ends up slower once reruns and investigation time are included.
Where throughput and consistency become linked
Throughput and consistency tend to become inseparable in multi-plate studies, larger cohorts, and longitudinal projects where samples arrive in waves. In those settings, turnaround time matters—but reruns still need to be minimized so the schedule remains predictable.
Why this matters in practice
Standardized washing supports more predictable plate handling and more stable QC outcomes. That predictability matters when results must be delivered on time, and it's one reason operational scalability becomes part of assay quality rather than a purely logistical concern.
When Automated Magnetic Washing Adds the Most Value
Higher-value use cases
Automated magnetic washing tends to pay off most in multi-plate Luminex cytokine studies, serum/plasma biomarker projects with moderate-to-large sample counts, and longitudinal programs where consistency across weeks or months matters. It's also helpful when samples are precious and reruns are expensive, or when the same workflow must be repeated across multiple operators or days.
Situations where automation becomes more important
Automation becomes more compelling when small biological effects must be distinguished from technical variation, when plate count grows beyond pilot scale, when timelines require predictable execution, or when reproducibility expectations move beyond exploratory screening.
A fit-for-purpose view
The point isn't that automated washing is always "better." The question is whether it materially improves the risk profile of your specific study—especially around reproducibility, rerun burden, and deliverability.
Related capability: If you need a validated, scalable workflow for multiplex cytokine panels in serum or plasma, see Serum/Plasma Cytokine Luminex Testing and broader options in Multiplex Cytokine Panel Services .
When Manual Washing Is Still Reasonable
Situations where manual washing may remain fit for purpose
Manual washing can be a reasonable choice for early feasibility work, very small pilot studies, limited plate counts, or exploratory projects where incremental gains in process consistency aren't yet critical.
Boundaries to keep in mind
What works in a small study doesn't always scale smoothly into production volume. As repetition increases, reliance on technician consistency becomes a larger source of drift, and the "acceptable" level of manual variation depends on study objectives, plate volume, and rerun tolerance.
A practical interpretation
Manual washing isn't inherently inadequate—it's simply more dependent on execution discipline and less inherently standardized than automated magnetic washing. Whether it's suitable depends on project complexity rather than abstract preference.
For pilot-to-production transitions, a service partner can help lock SOPs and QC gates. Explore tailored panel options in Custom Human Cytokine Assays .
Where Washing Method Starts to Affect Study Outcomes
Reproducibility
- Standardized washing can reduce avoidable process drift.
- More stable execution can help preserve smaller biological differences.
- The impact becomes more visible across repeated plates and longer workflows.
Failure risk
- Better wash consistency can reduce some handling-linked failure modes.
- Reduced rerun burden matters most when sample reserve is limited.
- In some studies, execution reliability is as important as raw assay sensitivity.
Throughput and deliverability
- Automation can support more predictable multi-plate scheduling.
- Predictable execution improves operational confidence.
- Deliverability matters more in later-stage or time-sensitive programs than in one-off pilot work.
What Automated Washing Does Not Replace
It does not replace fit-for-purpose assay design
- Poor panel composition still creates weak data.
- Wrong dilution strategy still affects reportability.
- Weak analytes remain weak even under more standardized washing.
It does not remove the need for QC review
- Bead count still matters.
- Plate consistency still matters.
- Control performance still matters.
- Batch review still matters in larger studies.
It does not eliminate other sources of variation
- Pre-analytical handling
- Matrix effects
- Reagent lots
- Instrument drift
- Study design imbalance
Common Misconceptions to Avoid
Automated washing guarantees better data
- It can reduce one source of variability, but it does not guarantee strong assay performance on its own.
Manual washing is always inadequate
- It can be fully acceptable in small or early-stage studies when the execution burden is limited.
Throughput is the only benefit of automation
- In many studies, the more important benefit is more stable repetitive execution.
Washing method does not affect reportability
- In practice, wash consistency can influence bead recovery, rerun burden, and data usability.
All projects benefit equally from automation
- The value depends on plate scale, sample value, reproducibility needs, and study objectives.
A Practical Decision Framework
Step 1: Define the study scale
- Small pilot
- Medium-sized biomarker study
- Large cohort
- Longitudinal or multi-batch program
Step 2: Define the execution sensitivity of the study
- Exploratory signal detection
- Translational prioritization
- Decision-support biomarker readout
- Cross-batch reproducibility requirement
Step 3: Assess operational risk
- Number of plates
- Sample value
- Rerun tolerance
- Timeline pressure
- Operator dependence
Step 4: Choose the least risky workflow
- Manual washing for smaller, lower-risk exploratory work
- Automated magnetic washing for studies requiring more stable, scalable execution
- Transition to automation when pilot workflows move toward production volume
Step 5: Align workflow choice with reporting expectations
- Stronger reproducibility expectations require stronger execution control
- Operational convenience should not outweigh data interpretability and deliverability
Helpful context for study planning: broader menu options are outlined in Multiplex Cytokine Panel Services and matrix-specific approaches in Serum/Plasma Cytokine Luminex Testing .
Manual vs Automated Magnetic Washing — Representative Parameters
Two modes of washing can be fit-for-purpose. Parameters below are representative; always follow the kit insert.
| Step | Manual magnetic washing (representative) | Automated magnetic washing (representative) |
|---|---|---|
| Magnet capture/settle | Place plate on magnetic block; hold ~60 s before aspiration | Program pre-soak/settle ~60 s before aspiration |
| Wash cycles per step | 2–3× (per capture/detection/SA-PE step) | 3× programmed cycles (kit-specific) |
| Wash volume | 100–200 µL per well per cycle | 100–200 µL per well per cycle (controlled dispense) |
| Aspiration | Tip angled to sidewall; avoid disturbing bead pellet | Height-controlled aspiration; low residual volume |
| Resuspension before read | Vortex/shake 30–60 s; gentle pipetting | Programmed mix + plate shaker; verify uniform suspension |
Sources: Agilent magnetic-bead wash app notes; MILLIPLEX kit inserts; Bio‑Rad/Bio‑Plex and R&D Systems user guides.
Troubleshooting Matrix for Wash-Linked Issues
| Symptom | Likely cause | Corrective action |
|---|---|---|
| Low bead counts | Over‑aspiration, inadequate resuspension, probe too low/high | Increase magnet hold time; adjust aspiration height; shake/vortex immediately before read; verify probe height and unclogged probes |
| High background | Incomplete washing; splash/carryover; degraded reagents | Ensure 2–3× washes after each binding step; verify dispense/aspiration rates; confirm buffer/reagent integrity |
| Well‑to‑well variability | Inconsistent timing/handling; variable residuals | Standardize soak/aspiration; consider automated cycles; ensure uniform shaking and resuspension |
Tip: Acquisition targets such as 100 beads per region (assay-dependent) help stabilize precision; low counts often trace back to resuspension and aspiration control, both of which benefit from consistent washing programs described in platform user guides.
Pre-Study Checklist
Scientific checklist
- Define the study objective.
- Decide whether execution consistency is likely to affect the main biological readouts.
- Determine whether smaller effect sizes require tighter process control.
Analytical checklist
- Confirm the assay workflow is appropriate for bead-based multiplex processing.
- Define how bead recovery and plate consistency will be reviewed.
- Align washing method with rerun logic and QC expectations.
Operational checklist
- Estimate plate volume and throughput needs.
- Decide whether manual execution is sustainable at the required scale.
- Standardize SOPs before study launch.
- Document why the chosen washing workflow is fit for purpose.
FAQ
Does automated magnetic washing improve every multiplex cytokine assay?
Not automatically. It is most valuable when execution consistency, rerun burden, and scale materially affect study quality.
Is manual washing acceptable for Luminex cytokine assays?
Yes. It can be fit for purpose in smaller or earlier-stage studies with limited operational complexity.
Does automation mainly improve throughput or reproducibility?
Both can improve. In many studies, the more important gain is reduced handling variability across repeated runs.
Can automated washing solve batch effects by itself?
No. It reduces one potential source of execution variability, but study design and QC strategy still determine cross-batch reliability.
When should a project move from manual to automated washing?
When plate count, sample value, rerun sensitivity, or reproducibility requirements make manual execution the riskier option.
What aspects of washing most affect bead retention and background control?
Magnet hold/settle time, aspiration height/flow, number of washes, and consistent resuspension. These parameters are standardized more tightly in automated programs—a core reason they support magnetic bead washing reproducibility.
How do I balance manual vs automated washing in a mixed study design?
Treat pilots as SOP‑shaping phases under manual control; lock parameters and QC gates, then transition to automation once plate counts and reproducibility expectations grow. If human studies require consistent longitudinal handling, automation typically decreases Luminex wash failure risk at scale.
References:
- Rountree W, et al. Sources of variability in Luminex bead‑based cytokine assays (J Immunol Methods, 2024). Available via the EQAPOL multi‑laboratory program: https://pmc.ncbi.nlm.nih.gov/articles/PMC11246216/
- Wu L, et al. Optimisation and standardisation of a multiplex immunoassay (2020): https://pmc.ncbi.nlm.nih.gov/articles/PMC7255915/
- Le Doare K, et al. Interlaboratory comparison of a multiplex immunoassay (2024): https://pmc.ncbi.nlm.nih.gov/articles/PMC11018077/
- Agilent. Automated Workflows for Luminex xMAP Assays (Application Note, 2021): https://www.agilent.com/cs/library/applications/automated-workflows-for-Luminex-xMAP-assays-5994-2625EN-agilent.pdf
- R&D Systems/Bio‑Techne. Luminex Assay Users Guide (2025): https://resources.bio-techne.com/bio-techne-assets/images/literature/br-luminex-assay-users-guide.pdf
- Bio‑Rad. Bio‑Plex System Hardware Instruction Manual (2021): https://www.bio-rad.com/sites/default/files/webroot/web/pdf/lsr/literature/4110018C.pdf
- MILLIPLEX Kit Insert (magnetic bead format; example with wash guidance, 2022): https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/270/710/protocol-mcytomag70k-mk.pdf

