Why endothelial dysfunction biomarkers matter in atherosclerosis research
Endothelial activation is the gateway to leukocyte recruitment and plaque biology. In the atherosclerotic artery, leukocytes are first captured and rolled along the endothelium by selectins, then transition to firm adhesion through immunoglobulin superfamily members (notably ICAM‑1 and VCAM‑1) before transmigrating into the intima. Circulating soluble forms—sICAM‑1, sVCAM‑1, E‑selectin, and P‑selectin—serve as research-friendly readouts of this activation cascade, offering a pragmatic window into vascular inflammation without direct tissue sampling. Reviews in vascular biology have consistently mapped these markers to the adhesion steps and lesion biology, including targeted summaries from Cardiovascular Research (2023) and Frontiers in Immunology (2024), which detail VCAM‑1's role in monocyte recruitment and E‑selectin's timing‑sensitive induction during endothelial activation.
These soluble adhesion molecules are best interpreted as complementary components of an endothelial dysfunction module. They are not diagnostic tests; instead, they are Research Use Only tools that contextualize vascular inflammation, complementing broader inflammation axes (e.g., chemokines or CRP) while staying focused on endothelial biology. Historically, prospective cohorts have shown associations between elevated soluble adhesion molecules and future coronary events, with the canonical prospective signal often traced to an early study in The Lancet (2001). More recent literature emphasizes mechanistic plausibility and disease‑context nuance rather than blanket prediction claims—an approach that aligns with cohort phenotyping and longitudinal change tracking.
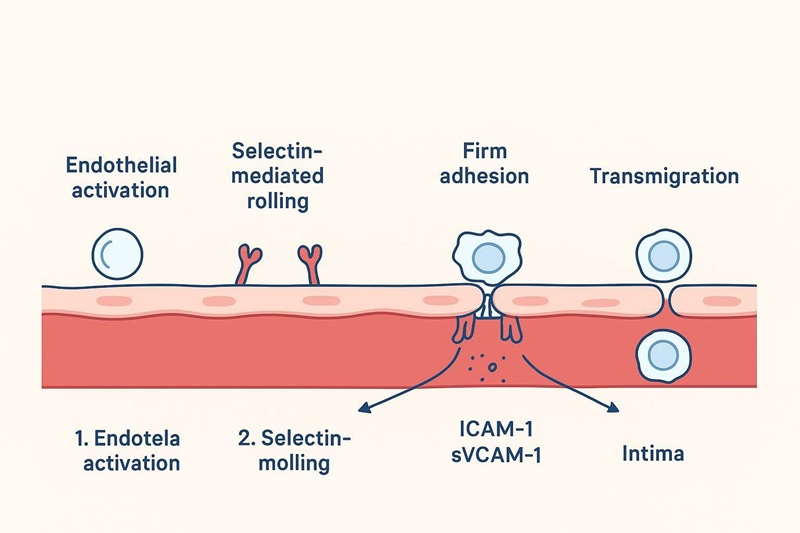 Biomarkers mapped to rolling, firm adhesion, and transmigration in atherosclerosis research.
Biomarkers mapped to rolling, firm adhesion, and transmigration in atherosclerosis research.
Endothelial dysfunction biomarkers in atherosclerosis: what to measure
sICAM-1 (soluble ICAM-1) for endothelial activation context
- Broad endothelial activation and vascular injury readout
- Firm-adhesion step proxy via ICAM-1 shedding
- Cohort-friendly inclusion in a cardiovascular biomarker module via a configurable multiplex panel or targeted assays as appropriate
sVCAM-1 (soluble VCAM-1) for vascular inflammation and recruitment
- Monocyte recruitment–aligned endothelial activation readout
- Atherosclerosis progression–aligned biomarker in published research, with cohort-dependent signal strength
- Core pairing logic with sICAM-1 for endothelial dysfunction panels
E-selectin for early activation and leukocyte rolling
- Endothelial activation–specific rolling signal
- Timing-sensitive marker for early stage or perturbation studies
- Multiplex feasibility within endothelial modules using the Luminex cytokine detection service
P-selectin for platelet–endothelium interface and thrombo-inflammation
- Platelet/endothelial activation axis and leukocyte tethering context
- Handling-sensitive marker requiring platelet activation control
- Species-appropriate studies can be supported with the Mouse Luminex multiplex assay panel when translating hypotheses in vivo
Choose biomarkers by research question (not one-size-fits-all)
Atherosclerosis stage emphasis
- Early endothelial activation: E-selectin-led module
- Established plaque inflammation: sVCAM-1 + sICAM-1 core
- Thrombo-inflammatory angle: P-selectin add-on
Study design emphasis
- Cross-sectional phenotyping: broad endothelial activation snapshot
- Longitudinal cohorts: drift-resistant plan and site harmonization
- Intervention studies: directional change markers and pre-specified response windows
Human cohorts vs mouse models
- Human confounding control and cohort scaling with standardized endothelial and inflammation modules
- Mouse mechanistic probing with species-matched panels via the Mouse Luminex multiplex assay panel
Quick comparison table: endothelial dysfunction biomarkers in atherosclerosis
| Biomarker | Best used for | Biological step | Best pairing | Handling sensitivity |
|---|---|---|---|---|
| sICAM-1 | Endothelial activation context | Firm adhesion | sVCAM-1 + minimal inflammation context | Medium |
| sVCAM-1 | Vascular inflammation/recruitment | Firm adhesion | sICAM-1 + chemokine axis (if justified) | Medium |
| E-selectin | Early activation/rolling emphasis | Rolling | sICAM-1/sVCAM-1 core | Medium–High |
| P-selectin | Thrombo-inflammation/platelet interface | Rolling + platelet-endothelium | sVCAM-1 core (hypothesis-driven) | High |
 A fast comparison of endothelial dysfunction biomarkers by mechanism, study fit, and handling sensitivity.
A fast comparison of endothelial dysfunction biomarkers by mechanism, study fit, and handling sensitivity.
Build a minimal vs expanded endothelial dysfunction biomarker module
Minimal module (high signal-to-noise)
- sICAM-1 + sVCAM-1 as the core endothelial activation pair
- One selectin chosen by hypothesis: E-selectin or P-selectin
- Deployment via multiplex or targeted assays depending on matrix, volume, and sensitivity plans
Expanded module (context without scope creep)
- Minimal inflammation context only (avoid duplicating large cytokine-only blocks)
- Optional oxidative stress context only when hypothesis-driven
- Custom expansion through the Serum & plasma cytokine assay for matrix-aligned, low-volume workflows
Interpretation pitfalls and confounders for soluble adhesion molecules
Biological confounding that can mimic endothelial dysfunction
- Systemic inflammation spillover vs vascular-specific activation
- Comorbidities and medication confounding (treat as research covariates)
- Acute stressors: exercise, circadian timing, recent procedures
Selectin-specific interpretation traps
- Platelet activation bias for P-selectin
- Timing sensitivity for E-selectin in acute perturbations
- Site-to-site collection variation as a false "signal"
Pre-analytical and matrix considerations (serum vs plasma)
Serum vs plasma decision points for endothelial biomarkers
Either matrix can be used in research, but consistency and pre-analytical control matter most. When selectins—especially P-selectin—are included, favor platelet-poor plasma (EDTA or citrate) with rapid processing and documented time-to-spin. Align anticoagulants across sites and timepoints to avoid spurious center effects. When feasible, define freeze–thaw limits (prefer ≤1) and flag hemolysis/lipemia/icterus.
- Anticoagulant consistency across sites and timepoints
- Platelet activation control when measuring selectins
- Matrix selection and SOP alignment can leverage the Serum & plasma cytokine assay for harmonized handling
Common pre-analytical pitfalls (checklist-style)
- Hemolysis/lipemia/icterus flagging and disposition rules
- Freeze–thaw minimization via aliquot strategy
- Shipping and site harmonization tie-in: standardize cold-chain, time-to-spin, and PPP options when selectins are included
 A pre-analytical checklist to reduce site-driven artifacts for endothelial dysfunction biomarkers.
A pre-analytical checklist to reduce site-driven artifacts for endothelial dysfunction biomarkers.
Measurement strategy: multiplex readout for endothelial dysfunction biomarkers
When multiplex makes sense
- Limited volume and multi-marker interpretation needs
- Cohort scaling and efficient plate workflows
- Practical multiplex execution through the Luminex cytokine detection service, with predefined range planning and controls
When to add targeted assays
- Handling-sensitive selectins requiring specialized controls
- Endpoint-critical markers needing distinct sensitivity profiles
- Keep platform deep-dives in cohort selection guides; in practice, combine multiplex screening with targeted follow-up when dynamic range or sensitivity constraints arise
Decision matrix table: when to include which marker
| Research scenario | Include first | Add next | Defer unless hypothesis-driven |
|---|---|---|---|
| Endothelial activation profiling in atherosclerosis | sICAM-1 + sVCAM-1 | E-selectin | Large unrelated cytokine blocks |
| Early activation/rolling emphasis | E-selectin | sICAM-1 | P-selectin without platelet controls |
| Thrombo-inflammation emphasis | P-selectin | sVCAM-1 | E-selectin if timing not controlled |
| Mouse model → human translation | Species-matched adhesion module | Minimal context markers | Human-only assays in mouse workflows |
Reporting checklist for endothelial dysfunction biomarker results
- Sample matrix and anticoagulant declared (serum/plasma harmonized)
- Pre-analytical flags reported (hemolysis/lipemia/icterus; freeze–thaw count)
- Plate/run metadata captured (batch identifiers; bridge samples if used)
- Units and transformations standardized (skew-aware reporting)
- Missingness annotated (sample QC vs quantification limits vs assay failure)
Related resource cluster links (non-overlapping intent)
- Cross-pathway architecture: How to design a cardiovascular biomarker panel spanning inflammation, adhesion, and oxidative stress
- Inflammation axis context without scope creep: When to use which inflammation biomarkers in cardiovascular research
- QC deep dive for acceptance rules: Multiplex assay QC acceptance criteria for cohort studies
FAQ
What are the best endothelial dysfunction biomarkers for atherosclerosis studies?
sICAM-1 and sVCAM-1 form the core of endothelial activation profiling, while E-selectin or P-selectin are added when rolling biology or thrombo-inflammation is central to the research question.
What is the difference between sICAM-1 and sVCAM-1?
Both reflect endothelial activation, but they map to overlapping yet distinct adhesion biology (ICAM-1 vs VCAM-1 axes). Measuring both often improves interpretability over using either alone—especially in longitudinal cohorts.
Should I measure E-selectin or P-selectin in atherosclerosis research?
Use E-selectin for endothelial activation and leukocyte rolling emphasis; choose P-selectin when platelet–endothelium interactions or thrombo-inflammatory biology is a key hypothesis and platelet activation can be tightly controlled.
Serum vs plasma: which is better for soluble adhesion molecule assays?
Either can work for research, but consistency and pre-analytical control matter most. When selectins (especially P-selectin) are included, platelet-poor plasma workflows with rapid processing are typically favored.
Can I run sICAM-1, sVCAM-1, and selectins in a single multiplex assay?
Yes. Multiplex formats can measure these together if range planning, matrix controls, and handling-sensitive markers (notably selectins) are managed with a predefined SOP.
Are soluble adhesion molecules specific to endothelial dysfunction?
They're informative for endothelial activation in research, but systemic inflammation and pre-analytical variation can influence levels. Interpretation should incorporate confounders and handling controls.
References:
- Blankenberg S, Rupprecht HJ, Bickel C, et al. Soluble adhesion molecules and prediction of coronary heart disease: a prospective study. The Lancet. 2001;358(9286):971–976. https://doi.org/10.1016/S0140-6736(01)06104-9
- Sitia S, Tomasoni L, Atzeni F, et al. From endothelial dysfunction to atherosclerosis. Autoimmunity Reviews. 2010;9(12):830–834. https://doi.org/10.1016/j.autrev.2010.07.016
- Rojas-Morales P, et al. E-selectin in vascular pathophysiology. Frontiers in Immunology. 2024;15:1401399. https://doi.org/10.3389/fimmu.2024.1401399
- Wang M, et al. Biomarkers of endothelial activation and dysfunction in cardiovascular diseases. Reviews in Cardiovascular Medicine. 2022;23(2):073. https://doi.org/10.31083/j.rcm2302073

